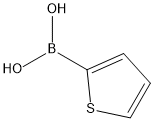
2-Thiopheneboronic acid synthesis
- Product Name:2-Thiopheneboronic acid
- CAS Number:6165-68-0
- Molecular formula:C4H5BO2S
- Molecular Weight:127.96
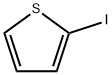
3437-95-4
243 suppliers
$6.00/5g

6165-68-0
385 suppliers
$14.00/5g
Yield:6165-68-0 99%
Reaction Conditions:
Stage #1: 2-Iodothiophenewith diisopropopylaminoborane;triethylamine;triphenylphosphine;palladium dichloride in tetrahydrofuran at 65; for 12 h;Inert atmosphere;Alcaraz-Vaultier borylation;
Stage #2: with methanol in tetrahydrofuran at 0;Inert atmosphere;Further stages;
Steps:
4.2. General procedure for the synthesis of boronic acids from the reaction of aryl iodides, bromides, or triflates with BH2N(iPr)2 in the presence of a palladium catalyst
General procedure: Triphenylphosphene (0.131 g, 0.5 mmol, 20 mol %), p-iodoanisol (0.585 g, 2.5 mmol), and triethylamine (1.78 mL, 12.5 mmol) were added to a 50 mL round-bottomed flask equipped with a sidearm, condenser, and stir bar. This solution was then degassed by alternating vacuum and argon three times. Palladium dichloride (0.023 g, 0.13 mmol, 5 mol %) was then added under positive argon pressure. After stirring at room temperature for 15 min, diisopropylaminoborane (5 mL, 1 M solution in THF, 5 mmol) was added and the reaction mixture was degassed again by alternating vacuum and argon three times. The reaction solution was then heated to reflux. After 12 h of reflux the reaction was cooled to 0 °C and 6 mL of methanol was added through the condenser slowly (Caution: exothermic reaction with evolution of hydrogen). After 15 min of stirring all the solvent was removed under reduced pressure to yield a black solid. This solid was dissolved with sodium hydroxide (3 M, 8 mL) and subsequently washed with hexanes (3×10 mL). The aqueous layer was then cooled to 0 °C (ice bath) and acidified to pH ≤1 with concentrated HCl, with the boronic acid usually precipitating out as a white solid. The aqueous fraction was then extracted with diethyl ether (3×10 mL). The organic fractions were combined, dried with magnesium sulfate and filtered. The solvent was then removed under reduced pressure yielding a white solid.
References:
Haddenham, Dustin;Bailey, Christopher L.;Vu, Chau;Nepomuceno, Gabby;Eagon, Scott;Pasumansky, Lubov;Singaram, Bakthan [Tetrahedron,2011,vol. 67,# 3,p. 576 - 583] Location in patent:experimental part
1003-09-4
466 suppliers
$10.00/25g

6165-68-0
385 suppliers
$14.00/5g
188290-36-0
1 suppliers
inquiry
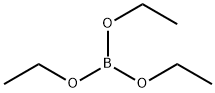
150-46-9
185 suppliers
$14.00/25mL

6165-68-0
385 suppliers
$14.00/5g

1003-09-4
466 suppliers
$10.00/25g
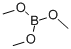
121-43-7
345 suppliers
$14.00/25mL

6165-68-0
385 suppliers
$14.00/5g

1003-09-4
466 suppliers
$10.00/25g

67-56-1
736 suppliers
$9.00/25ml
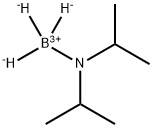
55124-35-1
23 suppliers
$60.00/100mg

6165-68-0
385 suppliers
$14.00/5g