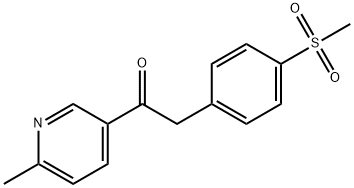
1-(6-Methylpyridin-3-yl)-2-(4-(methylsulfonyl)phenyl)ethanone synthesis
- Product Name: 1-(6-Methylpyridin-3-yl)-2-(4-(methylsulfonyl)phenyl)ethanone
- CAS Number:221615-75-4
- Molecular formula:C15H15NO3S
- Molecular Weight:289.35
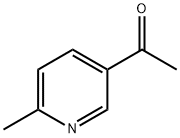
36357-38-7
174 suppliers
$15.00/250mg
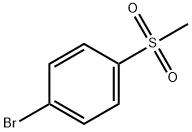
3466-32-8
194 suppliers
$8.00/1g

221615-75-4
267 suppliers
$8.00/250mg
Yield:221615-75-4 91%
Reaction Conditions:
with potassium phosphate;palladium(II) acetylacetonate;4,5-bis(diphenylphos4,5-bis(diphenylphosphino)-9,9-dimethylxanthenephino)-9,9-dimethylxanthene in 1-methyl-pyrrolidin-2-one at 100; for 18 h;Product distribution / selectivity;Inert atmosphere;
Steps:
9
Pd(acac)2 (6.1 mg, 0.02 mmol, 0.5 mol %) and Xantphos (23.2 mg, 0.04 mmol, 1 mol o) are introduced into a flared flask provided with coolant. 4-bromophenylmethylsulfone of formula (III, XBr) (1.17 g, 5 mmol), acetylpicoline of formula (II) (541 mg, 4 mmol) and K3PO4 (2.55 g, 12.0 mmol, 3 eq) are added thereto. Once the argon atmosphere has been stabilized with vacuum-argon cycles, anhydrous and degassed NMP (15 ml) is added with a syringe. The mixture is then kept stirred under stirring in an argon atmosphere for 18 h at 100° C. The conversion is quantitative. The reaction mixture is diluted with a saturated solution of NaHCO3 (50 mL) and extracted with AcOEt (4×50 mL). The combined organic phases were washed with an aqueous solution saturated with NaHCO3 (30 mL), anhydrified on MgSO4 and concentrated in a vacuum. The residue was purified by silica gel chromatography using AcOEt/cyclohexane as eluent in a gradient from 5:5 to 10:0. 1.05 g product were obtained, for a molar yield of 91% as a white crystalline solid.
References:
Castellin, Andrea;Stabile, Paolo;Fontana, Francesco;Lucchi, Ottorino De;Caporale, Andrea;Tartaggia, Stefano US2012/232281, 2012, A1 Location in patent:Page/Page column 7-8
221615-72-1
93 suppliers
$30.00/250mg

221615-75-4
267 suppliers
$8.00/250mg
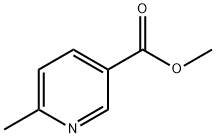
5470-70-2
513 suppliers
$5.00/5g
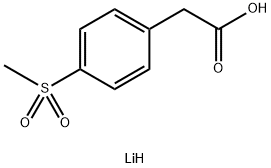
1421227-96-4
11 suppliers
inquiry
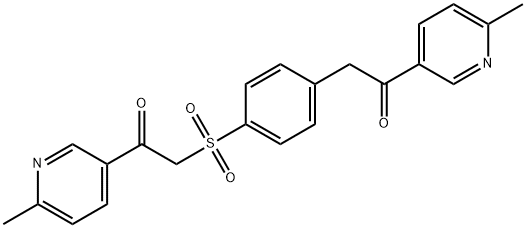
1421227-97-5
32 suppliers
inquiry

221615-75-4
267 suppliers
$8.00/250mg

5470-70-2
513 suppliers
$5.00/5g
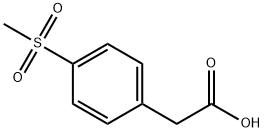
90536-66-6
312 suppliers
$6.00/1g

221615-75-4
267 suppliers
$8.00/250mg

5470-70-2
513 suppliers
$5.00/5g

90536-66-6
312 suppliers
$6.00/1g

1421227-97-5
32 suppliers
inquiry

221615-75-4
267 suppliers
$8.00/250mg