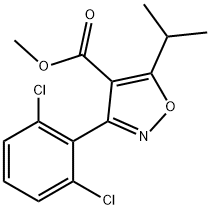
3-(2,6-DICHLORO-PHENYL)-5-ISOPROPYL-ISOXAZOLE-4-CARBOXYLIC ACID METHYL ESTER synthesis
- Product Name:3-(2,6-DICHLORO-PHENYL)-5-ISOPROPYL-ISOXAZOLE-4-CARBOXYLIC ACID METHYL ESTER
- CAS Number:278597-28-7
- Molecular formula:C14H13Cl2NO3
- Molecular Weight:314.16
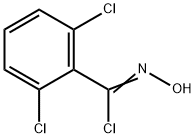
6579-27-7
66 suppliers
$45.00/100mg

42558-54-3
384 suppliers
$10.00/5g

278597-28-7
33 suppliers
$275.00/500mg
Yield:278597-28-7 69%
Reaction Conditions:
Stage #1: Methyl 4-methyl-3-oxopentanoatewith sodium methoxide in tetrahydrofuran;methanol at 0;
Stage #2: 2,6-dichloro-N-hydroxylbenzene-1-carbonimidoyl chloride in tetrahydrofuran;methanol at 20;
Steps:
b b) Methyl 3-(2,6-dichlorophenyl)-5-isopropylisoxazole-4-carboxylate.
A solution of 2, 6-DICHLOROBENZALDEHYDE oxime (19. 8g, 0.104 mol) in N, N-dimethyl formamide (80 mL) was placed in an ambient temperature water bath and was treated with N-CHLOROSUCCINIMIDE (13.9g, 0.104 mole). Following dissolution, an exotherm was observed along with a color change to dark yellow. The reaction was stirred an additional hour then the contents were then poured into water (200 mL) and the product extracted with diethyl ether (300mL). The ethereal layer was washed with water (3x 100 mL) and brine (50mL), then dried over anhydrous magnesium sulfate. After filtering, the solvent was removed in vacuo to yield a yellow oil which was used in the next step without further purification. Separately, a solution of methyl isobutyryl acetate (18g, 0.125 mol) in tetrahydrofuran (25 mL) at 0°C was treated with a solution of sodium methoxide (250 mL, 0.5 M in methanol). A solution of the above crude 2, 6-DICHLORO-N-HYDROXYBENZENECARBOXIMIDOYL chloride in tetrahydrofuran (80 mL) was then added dropwise. After stirring at ambient temperature for 16h the solvent was removed in vacuo. The residue was triturated with water (250 mL) and the resulting solids filtered and washed with water. Yield = 22.7 g. (69%) of methyl 3- (2, 6-dichlorophenyl)-5-isopropylisoxazole-4-carboxylate.
References:
WO2004/48349,2004,A1 Location in patent:Page 46

42558-54-3
384 suppliers
$10.00/5g

6579-27-7
66 suppliers
$45.00/100mg

278597-28-7
33 suppliers
$275.00/500mg

83-38-5
444 suppliers
$6.00/10g

278597-28-7
33 suppliers
$275.00/500mg

42558-54-3
384 suppliers
$10.00/5g

25185-95-9
125 suppliers
$24.89/5g

278597-28-7
33 suppliers
$275.00/500mg

25185-95-9
125 suppliers
$24.89/5g

278597-28-7
33 suppliers
$275.00/500mg