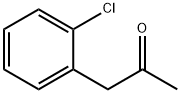
3-(2-Chlorophenyl)butan-2-one synthesis
- Product Name:3-(2-Chlorophenyl)butan-2-one
- CAS Number:21905-96-4
- Molecular formula:C10H11ClO
- Molecular Weight:182.65
Yield:21905-96-4 69%
Reaction Conditions:
Stage #1: 2-chlorophenylacetonewith benzyltrimethylammonium chloride;sodium hydroxide in water at 0; for 0.166667 h;
Stage #2: methyl iodide in water at 0 - 20; for 2 h;
Steps:
2.i Step-i: 3-(2-chlorophenyl)butan-2-one (2a)
Step-i: 3-(2-chlorophenyl)butan-2-one (2a) To a 25 mL round bottom flask, was added l-(2-chlorophenyl)propan-2-one (2 g, 0.0118 mol) and cooled to 0 °C. To the same flask, 50 % aqueous sodium hydroxide (10 mL) and benzyltrimethylammonium chloride (0.22 g, 0.0011 mol) were added. The reaction mixture was stirred at 0 °C for 10 min. To the reaction mixture, methyl iodide (2.02 g, 0.0142 mol) was added and stirred at room temperature for 2 h. The reaction mixture was diluted with water and extracted with dichloromethane. The organic layer was separated, washed with brine and dried over anhydrous sodium sulfate. The organic layer was evaporated under reduced pressure to get the crude product. The crude product was purified by column chromatography over silica gel (60-120) using 3 % ethyl acetate in hexane as eluent to get the titled compound [1.5 g, 69 %].1H NMR (300 MHz, CDC13): δ 7.43-7.40 (m, 1H), 7.27-7.14 (m, 3H), 4.30 (q, 1H), 2.08 (s, 3H), 1.39 (d, 3H).
References:
WO2015/83130,2015,A1 Location in patent:Page/Page column 31-32
82234-99-9
0 suppliers
inquiry

21905-96-4
4 suppliers
inquiry

89-98-5
637 suppliers
$15.00/25g

21905-96-4
4 suppliers
inquiry