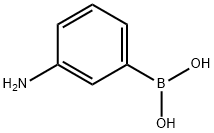
3-Aminobenzeneboronic acid synthesis
- Product Name:3-Aminobenzeneboronic acid
- CAS Number:30418-59-8
- Molecular formula:C6H8BNO2
- Molecular Weight:136.94

13331-27-6
350 suppliers
$5.00/1g

30418-59-8
248 suppliers
$6.00/1g
Yield:30418-59-8 83%
Reaction Conditions:
Stage #1:m-nitrobenzene boronic acid with hydrogenchloride;1,1,1,3',3',3'-hexafluoro-propanol;iron in water at 20; for 0.5 h;
Stage #2: with sodium hydrogencarbonate in waterchemoselective reaction;
Steps:
2. General Procedure for the Reduction of Nitro Compounds
General procedure: The nitro compound (1 equiv), HFIP (10 equiv), Fe powder (5 equiv) were mixed in a tube. Then 2 N HCl aqueous solutions was added to the reaction mixture. After stirring at room temperature for 30 min, the reaction mixture was neutralized with sat. NaHCO3 (aq.) and extracted with EtOAc three times. The combined organic layers were washed with brine, dried over Na2SO4, filtered, and concentrated under reduced pressure. The resulting crude product was then purified by column chromatography on silica gel to furnish the desired amine product.
References:
Chen, Xu-Ling;Ai, Bai-Ru;Dong, Yu;Zhang, Xiao-Mei;Wang, Ji-Yu [Tetrahedron Letters,2017,vol. 58,# 37,p. 3646 - 3649] Location in patent:supporting information
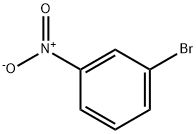
585-79-5
7 suppliers
$15.00/1g
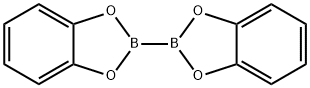
13826-27-2
133 suppliers
$5.00/250mg

30418-59-8
248 suppliers
$6.00/1g
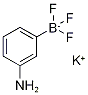
1160186-73-1
20 suppliers
$85.00/500mg
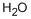
7732-18-5
461 suppliers
$12.69/100ml

30418-59-8
248 suppliers
$6.00/1g
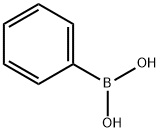
98-80-6
714 suppliers
$5.00/5g

30418-59-8
248 suppliers
$6.00/1g
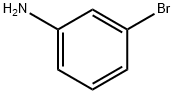
591-19-5
322 suppliers
$10.00/10g

30418-59-8
248 suppliers
$6.00/1g