
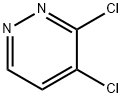
3-CHLORO-6-PHENOXYPYRIDAZINE synthesis
- Product Name:3-CHLORO-6-PHENOXYPYRIDAZINE
- CAS Number:1490-44-4
- Molecular formula:C10H7ClN2O
- Molecular Weight:206.63

141-30-0
396 suppliers
$6.00/10g
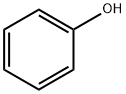
108-95-2
722 suppliers
$14.00/25g

1490-44-4
20 suppliers
$45.00/10mg
Yield:1490-44-4 93%
Reaction Conditions:
with copper(l) iodide;potassium carbonate in dimethyl sulfoxide at 90;
Steps:
II.85.1
A mixture of 3,6-dichloropyridazine (1.0 g, 5.23 mmol), phenol (0.50 g, 5.31 mmol), potassium carbonate (2.90 g, 20.98 mmol), and copper(I) iodide (0.59 g, 3.09 mmol) in dimethylsulfoxide (3.6 mL, 1.45 M) was heated to 90° C. overnight. After this time, the reaction was cooled to 25° C. and then poured into a 2N aqueous hydrochloric acid solution (75 mL) rinsing with water. The resulting mixture was filtered through filter paper and diluted with a saturated aqueous sodium chloride solution followed by extraction with ethyl acetate (150 mL). The organics were washed with a saturated aqueous sodium chloride solution (1×75 mL), dried over magnesium sulfate, filtered and concentrated in vacuo. Silica gel column chromatography (AnaLogix, 80 g, 5-24% ethyl acetate/hexanes) afforded 3-chloro-6-phenoxy-pyridazine (1.01 g, 93%) as a yellow solid. 1H-NMR (300 MHz, DMSO-d6) δ ppm 7.14-7.35 (m, 3H), 7.47 (t, J=7.5 Hz, 2H), 7.59 (d, J=9.2 Hz, 1H), 7.96 (d, J=9.2 Hz, 1H).
References:
US2009/264434,2009,A1 Location in patent:Page/Page column 119
1677-80-1
72 suppliers
$45.00/10mg

108-95-2
722 suppliers
$14.00/25g

1490-44-4
20 suppliers
$45.00/10mg