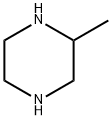
3-METHYL-1-(TOLUENE-4-SULFONYL)-PIPERAZINE synthesis
- Product Name:3-METHYL-1-(TOLUENE-4-SULFONYL)-PIPERAZINE
- CAS Number:178624-90-3
- Molecular formula:C12H18N2O2S
- Molecular Weight:254.35
Yield:178624-90-3 97%
Reaction Conditions:
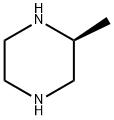
Stage #1: (S)-2-methylpiperazinewith triethylamine in dichloromethane at 0; for 0.0833333 h;
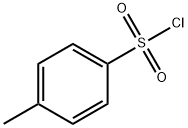
Stage #2: p-toluenesulfonyl chloride in dichloromethane at 0 - 20; for 5 h;
Steps:
4.2.6 (2S)-1,2-Dimethyl-4-(para-toluenesulfonyl)piperazine 11
Triethylamine (190μl, 1.37mmol) was added to a solution of commercially available (S)-(+)-2-methylpiperazine (125mg, 1.25mmol) in CH2Cl2 (7mL) at 0°C and stirred for 5min. p-Toluenesulfonyl chloride (238mg, 1.25mmol) was then added and the mixture was stirred for 1h at 0°C before being allowed to warm to room temperature and stirred for a further 4h. Water was then added and the aqueous layer was separated and extracted with CH2Cl2, before being washed sequentially with 1M HCl (50mL), water (50mL), saturated aqueous NaHCO3 solution (50mL) and saturated aqueous sodium chloride solution (50mL). The organic layer was separated, dried (Na2SO4), filtered and concentrated in vacuo to afford tosyl piperazine, (3S)-3-methyl-1-[(4-methylbenzene)sulfonyl]piperazine (310mg, 97%) as a white solid. The product could be used without further purification; mp 138.5-141°C (acetone); [α]23D=+38.9[α]D23=+38.9 (c 0.93, CHCl3); νmax (ATR)/cm-1 3352, 2971, 2922, 2863, 2821, 1605, 1452, 1325, 1165, 1133, 1121, 997, 914, 861, 811, 754s, 655; δH (500MHz, DMSO-d6) 7.61-7.59 (2H, m, 2×ArH), 7.47-7.44 (2H, m, 2×ArH), 3.41-3.37 (2H, m, CH2NTs), 2.85 (1H, dt, J 12.1 and 2.8, CH2NTs), 2.68-2.60 (2H, m, 3-H and CHHN(H)), 2.41 (3H, s, ArCH3), 2.06 (1H, td, J 11.3 and 3.1, CHHN(H)), 1.70 (1H, t, J 10.8, CHHN(H)), 0.90 (3H, d, J 6.4, 3-(CH3)); δC (126MHz, DMSO-d6) 143.4, 131.9, 129.7, 127.5, 52.3, 49.5, 45.9, 44.3, 20.9, 18.8; m/z (CI+) 255 (MH+, 100%), 99 (M+ -Ts, 10); HRMS (CI+) MH+ calculated for C12H19N2O2S 255.1167, observed 255.1164.
References:
Armstrong, Alan;Pullin, Robert D. C.;Jenner, Chloe R.;Foo, Klement;White, Andrew J. P.;Scutt, James N. [Tetrahedron Asymmetry,2014,vol. 25,# 1,p. 74 - 86]