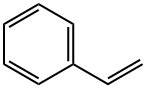
4,4,4-Trifluoro-1-phenylbutan-1-one synthesis
- Product Name:4,4,4-Trifluoro-1-phenylbutan-1-one
- CAS Number:713-02-0
- Molecular formula:C10H9F3O
- Molecular Weight:202.17
Yield:713-02-0 337 mg
Reaction Conditions:
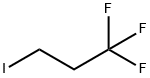
Stage #1: 1,1,1-trifluoro-3-iodopropanewith iodine;magnesium in diethyl ether; for 0.5 h;Inert atmosphere;Reflux;
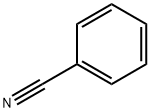
Stage #2: benzonitrilewith copper(I) bromide in tetrahydrofuran;diethyl ether at 0 - 50; for 2 h;Inert atmosphere;Grignard Reaction;
Steps:
General procedure A for the formation of 1,1,1-trifluoroalkanones 5
General procedure: A dry 2-necked round-bottomed flask attached to a condenser was filled with magnesium (8 mmol, 2equiv) under argon and stirred for 30 minutes. An iodine crystal was added. The mixture was warmedgently and stirred vigorously for 10 minutes. Dry diethyl ether (10 mL) was added followed by thedropwise addition of 1,1,1-trifluoro-4-iodo-butane (4 mmol, 2 equiv) at such a rate as to maintain agentle reflux. After the addition was complete, the mixture was stirred at reflux for an additional30 minutes.This solution of 4,4,4-trifluorobutylmagnesium iodide (10 mL, 2 equiv) was added to a solution of thecorresponding benzonitrile (4 mmol, 1 equiv) and CuBr (0.2 mmol, 5.0 mol %) in dry THF (10 mL) at0 °C under argon. The reaction was heated to 50 °C for 2 h. Upon completion the reaction was cooledto room temperature and quenched with 2M HCl (5 mL). The aqueous layer was extracted with ethylacetate (3 × 20 mL). The organic layers were combined, dried (MgSO4), filtered, and concentrated. Theresulting residue was purified by automated flash column chromatography (40 g silica, 0-100%heptane/ethyl acetate). In some cases further purification by reversed-phase column chromatographywas required (12 g silica, 0-100% water/acetonitrile).
References:
Lansbergen, Beatrice;Meister, Catherine S.;McLeod, Michael C. [Beilstein Journal of Organic Chemistry,2021,vol. 17,p. 404 - 409] Location in patent:supporting information
![Benzene, [1-[(triethylsilyl)oxy]ethenyl]-](/CAS/20210305/GIF/17718-70-6.gif)