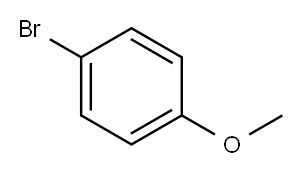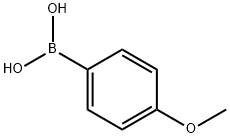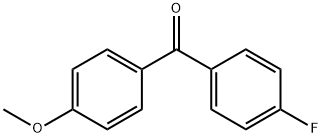
4-Fluoro-4'-methoxybenzophenone synthesis
- Product Name:4-Fluoro-4'-methoxybenzophenone
- CAS Number:345-89-1
- Molecular formula:C14H11FO2
- Molecular Weight:230.23
Yield: 85%
Reaction Conditions:
Stage #1:1-bromo-4-methoxy-benzene with n-butyllithium in tetrahydrofuran;hexane at -78; for 0.5 h;
Stage #2:4-fluorobenzaldehyde in tetrahydrofuran;hexane at -78 - 20; for 1 h;
Stage #3: with iodine;potassium carbonate in tert-butyl alcohol for 3 h;Reflux;
Steps:
4.2. Typical experimental procedure for one-pot conversion of aromatic bromides into ketones
General procedure: n-Butyllithium (1.67 M solution in hexane, 1.32 mL, 2.2 mmol) was added dropwise to a solution of 1-bromo-4-methylbenzene (342 mg 2.0 mmol) in THF (3 mL) at -78 °C for 30 min. Then, 4-fluorobenzaldehyde (261 mg, 2.1 mmol) was added to the mixture at -78 °C and the obtained mixture was stirred at room temperature for 1 h. Then, after removal of the solvent, I2 (812 mg,3.2 mmol), K2CO3 (829 mg, 6.0 mmol), and t-BuOH (3 mL) were added and the obtained mixture was stirred for 3 h at refluxing conditions. The reaction mixture was quenched with satd aq Na2SO3 (5 mL) and was extracted with CHCl3 (3×20 mL). The organic layer was washed with brine and dried over Na2SO4 to provide 4-fluoro-4'-methylbenzophenone in 98% yield with high purity. If necessary, the product was purified by a short flash columnchromatography (silica gel; hexane/CHCl3=1:3) to give pure 4-fluoro-4'-methylbenzophenone as a colorless solid.
References:
Ushijima, Sousuke;Dohi, Souya;Moriyama, Katsuhiko;Togo, Hideo [Tetrahedron,2012,vol. 68,# 5,p. 1436 - 1442] Location in patent:experimental part
201230-82-2
1 suppliers
inquiry
696-62-8
355 suppliers
$5.00/1g
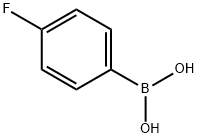
1765-93-1
558 suppliers
$9.00/1g

345-89-1
192 suppliers
$10.00/1g
![Ethanamine, N-[(3,5-dichlorophenyl)methylene]-2,2-diethoxy-](/CAS/20210305/GIF/1000210-73-0.gif)
1000210-73-0
0 suppliers
inquiry

345-89-1
192 suppliers
$10.00/1g