
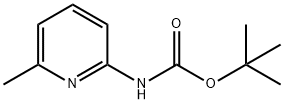
(6-TERT-BUTOXYCARBONYLAMINO-PYRIDIN-2-YL)-ACETIC ACID ETHYL ESTER synthesis
- Product Name:(6-TERT-BUTOXYCARBONYLAMINO-PYRIDIN-2-YL)-ACETIC ACID ETHYL ESTER
- CAS Number:408365-87-7
- Molecular formula:C14H20N2O4
- Molecular Weight:280.32
90101-22-7
72 suppliers
$42.60/1g

105-58-8
448 suppliers
$17.00/25g

408365-87-7
47 suppliers
$40.00/100mg
Yield:408365-87-7 100%
Reaction Conditions:
Stage #1: 2-(tert-butoxycarbonylamino)-6-picolinewith lithium diisopropyl amide in tetrahydrofuran;n-heptane at -78; for 0.5 h;
Stage #2: Diethyl carbonate in tetrahydrofuran;n-heptane at -78 - 0; for 2.16667 h;
Steps:
2.B; 4.H
2B. (β-tei't-Butoxycarbonylamino-pyridin-l-yl) acetic acid ethyl ester; To a solution of title compound 2A, (6-methyl-pyridin-2-yl) carbamic acid t?T-butyl ester, (0.97 g, 4.66 mmol) in anhydrous THF at -780C is added LDA (1.8 M solution in heptanes; 4 eq, 18.6 mmol) and the mixture is stirred for 30 min. Diethyl carbonate (2 eq, 9.32 mmol) is added and the mixture is stirred at -780C for a further 10 min. The mixture is allowed to warm to O0C and stirring continued for 2 h. The reaction is quenched with saturated NH4Cl solution. The solution is extracted with ethyl acetate. The combined organic phase is dried (MgSO4) and concentrated in vacuo. The crude product is purified by column chromatography with heptane/ ethyl acetate (gradient 98:2 - 95:5) to give the title compound (1.36 g, >100% repeated drying can not remove all traces of solvent)
References:
WO2006/50908,2006,A1 Location in patent:Page/Page column 32; 39-40

1824-81-3
474 suppliers
$8.00/25g

408365-87-7
47 suppliers
$40.00/100mg

24424-99-5
822 suppliers
$13.50/25G

408365-87-7
47 suppliers
$40.00/100mg