
6-CHLOROMETHYL-4-(4-METHOXY-PHENYL)-2-OXO-1,2,3,4-TETRAHYDRO-PYRIMIDINE-5-CARBOXYLIC ACID ETHYL ESTER synthesis
- Product Name:6-CHLOROMETHYL-4-(4-METHOXY-PHENYL)-2-OXO-1,2,3,4-TETRAHYDRO-PYRIMIDINE-5-CARBOXYLIC ACID ETHYL ESTER
- CAS Number:475042-38-7
- Molecular formula:C15H17ClN2O4
- Molecular Weight:324.76

638-07-3
470 suppliers
$5.00/5G

123-11-5
838 suppliers
$10.00/10g
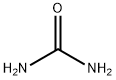
57-13-6
700 suppliers
$5.00/5g

475042-38-7
18 suppliers
$59.00/5mg
Yield:475042-38-7 98%
Reaction Conditions:
with hydrogenchloride in water at 100; for 2 h;Biginelli Pyrimidone Synthesis;
Steps:
4.2.1. General procedure for the synthesis of compounds 2a-hand 13
General procedure: The literature procedure was followed.24 To a two-neckedround-bottom flask, the appropriated aldehyde (10 mmol), ethyl 4-chloroacetoacetate or ethylacetoacetate (10 mmol), urea (1.2 g,20 mmol) and 5 drops of concentrated HCl were added. The reaction mixture was stirred at 100 °C for the time required for the consumption of the starting materials, which was verified by TLC. Afterthis time, the reaction mixture was poured into crushed ice and water. The precipitate was filtered off and dried. The compounds were used without further purification.
References:
Barbosa, Flavio A.R.;Canto, R?mulo F.S.;Saba, Sumbal;Rafique, Jamal;Braga, Antonio L. [Bioorganic and Medicinal Chemistry,2016,vol. 24,# 22,p. 5762 - 5770]