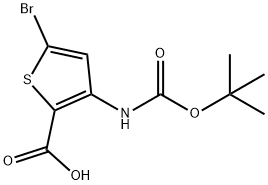
5-BroMo-3-tert-butoxycarbonylaMino-thiophene-2-carboxylic acid synthesis
- Product Name:5-BroMo-3-tert-butoxycarbonylaMino-thiophene-2-carboxylic acid
- CAS Number:494833-77-1
- Molecular formula:C10H12BrNO4S
- Molecular Weight:322.18
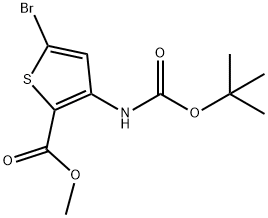
946604-99-5
14 suppliers
$186.00/1g

494833-77-1
19 suppliers
$65.00/100mg
Yield:494833-77-1 100%
Reaction Conditions:
with water;potassium hydroxide in methanol at 70; for 1 h;
Steps:
3.1.2 3.1.2) Synthesis of 5-bromo-3-((tert-butoxycarbonyl)amino)thiophene-2-carboxylic acid (compound 15)
To a stirred solution of compound 14 (3.30 g, 9.82 mmol) in MeOH (90 mL) was added 10% KOH aq (30 mL), the mixture was heated to 70°C for 1 h, LC-MS showed that the start materials was consumed up, stopped the reaction, cooled to rt, the pH was adjusted to 2-3 by adding 10% HC1, then concentrated to remove MeOH, the residue was extracted with DCM (250 mL*2), the combined organic layer was washed with brine, concentrated to give compound 15(3.30 g, 100% yield) as a yellow solid. MS (ESI) (M/Z): [M-H]- =319.9,321.9.
References:
WO2018/217439,2018,A1 Location in patent:Page/Page column 29-30
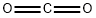
124-38-9
122 suppliers
$214.00/14L

494833-77-1
19 suppliers
$65.00/100mg
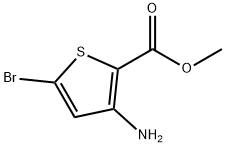
107818-55-3
82 suppliers
$27.00/100mg

494833-77-1
19 suppliers
$65.00/100mg

24424-99-5
824 suppliers
$13.50/25G

494833-77-1
19 suppliers
$65.00/100mg