
5-(2-TRIFLUOROMETHYL-PHENYL)-CYCLOHEXANE-1,3-DIONE synthesis
- Product Name:5-(2-TRIFLUOROMETHYL-PHENYL)-CYCLOHEXANE-1,3-DIONE
- CAS Number:55579-73-2
- Molecular formula:C13H11F3O2
- Molecular Weight:256.22

76293-37-3
16 suppliers
$45.00/10mg
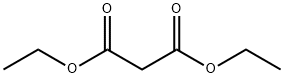
105-53-3
733 suppliers
$5.00/25g

55579-73-2
39 suppliers
$33.00/100mg
Yield:-
Reaction Conditions:
Stage #1: 2-trifluoromethylbenzalacetone;diethyl malonatewith sodium ethanolate in ethanol; for 12 h;
Stage #2: with sodium hydroxide in ethanol;water; for 12 h;
Stage #3: with hydrogenchloride in diethyl ether;water;
Steps:
4.2. General procedure to prepare cyclohexane 1,3-dione (CHD) analogs7
General procedure: All aldehydes were previously prepared or purchased. Aldehyde 1 (5.0 mmol) was dissolved in THF (20 mL), and 1-(triphenylphosphoranylidene)-2-propanone (1.60 g, 5.0 mmol) was added. The mixture was allowed to stir at room temperature for 12 h. Solvent was removed under vacuum, and the residue was purified by flash column chromatography to provide 2, which was used directly in the next step, in almost quantitative yield.Compound 2 (2.0 mmol) and diethyl malonate (2.0 mmol) were dissolved in EtOH (5 mL), and a NaOEt solution (21 wt % in ethanol; 1.50 mL, 4.0 mmol) was added. After being stirred for 12 h, 3 N aq NaOH (20 mL) was added, and stirring was continued for another 12 h. Aqueous 3 N HCl was added until the solution turned cloudy. Ether was used to extract the cloudy mixture until the aqueous layer was clear. The combined organic portions were combined and allowed to stir for 4 h until decarboxylation was complete. Heating might be required in some cases when decarboxylation was not complete at room temperature. The solvent was evaporated, and the crude product was then purified by recrystallization or flash chromatography. The CHD product was typically a white solid, obtained in about an 80% yield, and had an Rf = 0.6 (dichloromethane/methanol = 8:1).
References:
Zhang, Wei;Benmohamed, Radhia;Arvanites, Anthony C.;Morimoto, Richard I.;Ferrante, Robert J.;Kirsch, Donald R.;Silverman, Richard B. [Bioorganic and Medicinal Chemistry,2012,vol. 20,# 2,p. 1029 - 1045] Location in patent:experimental part
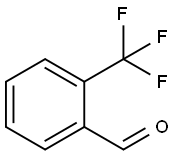
447-61-0
314 suppliers
$9.00/10g

55579-73-2
39 suppliers
$33.00/100mg

76293-37-3
16 suppliers
$45.00/10mg

55579-73-2
39 suppliers
$33.00/100mg