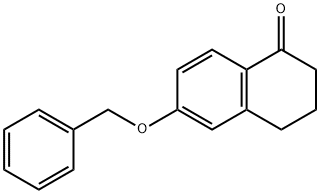
6-(benzyloxy)-3,4-dihydronaphthalen-1(2H)-one synthesis
- Product Name:6-(benzyloxy)-3,4-dihydronaphthalen-1(2H)-one
- CAS Number:32263-70-0
- Molecular formula:C17H16O2
- Molecular Weight:252.31
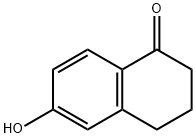
3470-50-6
225 suppliers
$6.00/1g
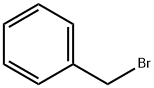
100-39-0
434 suppliers
$10.00/10 g

32263-70-0
114 suppliers
$18.00/100mg
Yield:32263-70-0 97%
Reaction Conditions:
with potassium carbonate in acetone for 6 h;Reflux;
Steps:
3. Synthesis of α-tetralone derivatives 6a-o
General procedure: 6-Hydroxy-1-tetralone (0.3 g, 1.85 mmol) was suspended in acetone (15 mL) containing K2CO3 (3.70 mmol). The reaction was treated with an appropriately substituted arylalkyl bromide (2.035 mmol) and heated under reflux for 6 h. The reaction progress was monitored using silica gel TLC with ethyl acetate:petroleum ether (1:2) as mobile phase. Upon completion, the reaction was filtered through a pad of celite and concentrated in vacuo. The crude thus obtained was purified by recrystallization from cyclohexane.
References:
Legoabe, Lesetja J.;Petzer, Anél;Petzer, Jacobus P. [Bioorganic and Medicinal Chemistry Letters,2014,vol. 24,# 12,p. 2758 - 2763] Location in patent:supporting information

3470-50-6
225 suppliers
$6.00/1g
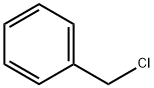
100-44-7
647 suppliers
$13.50/250G

32263-70-0
114 suppliers
$18.00/100mg
1078-19-9
376 suppliers
$6.00/5g

32263-70-0
114 suppliers
$18.00/100mg