
N-(tetrahydro-2H-pyran-4-yl)-4-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)benzamide synthesis
- Product Name:N-(tetrahydro-2H-pyran-4-yl)-4-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)benzamide
- CAS Number:656239-37-1
- Molecular formula:C18H26BNO4
- Molecular Weight:331.21
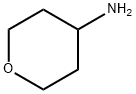
38041-19-9
321 suppliers
$6.00/1g
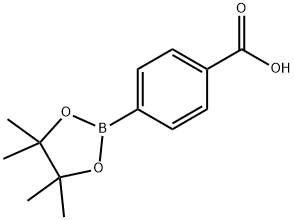
180516-87-4
227 suppliers
$8.00/1g

656239-37-1
23 suppliers
inquiry
Yield:656239-37-1 18%
Reaction Conditions:
Stage #1: 4-carboxyphenylboronic acid pinacol esterwith thionyl chloride for 2 h;Heating / reflux;
Stage #2: 4-aminotetrahydropyranwith triethylamine in dichloromethane;toluene at 10 - 20; for 48 h;
Steps:
13 Intermediate 13: [ 4-(4,4. 5. 5-TETRAMETHYL-F1, 3, 21DIOXABOROLAN-2-VL)-N- (TETRAHYDRO-PYRAN-] [4-VL)-BENZAMIDE]
[4- (4,] 4,5, 5-Tetramethyl- [1, 3,2] [DIOXABOROLAN-2-YL)-BENZOIC] acid (70. 16g, 0.28 mol) was treated with [SOC12] (2 [VOL.)] and the reaction mixture was stirred to reflux for 2 hours. After evaporation, the residue was diluted in toluene and poured into a solution at [10°C] of [TETRAHYDRO-PYRAN-4-YLAMINE] (34.34g, 0.339) and triethylamine (79 mL, 0.57 mol) in CH2CI2. The reaction mixture was stirred at room temperature for 2 days and water (490 mL) was added to give a precipitate which was filtered off and washed with ethyl acetate. After purification by flash chromatography using [CH2CI2/MEOH] (95: 5). The title compound was obtained as a [SOLID (17.] 02g, 18%) ;'H NMR (400 MHz, CDCl3, ppm) 8 : 7. 85 (d, 2H), 7.72 (d, 2H), 5.98 (m, [1H),] 4.20 (s, 1H), 3.99 (m, 2H), 3.35 (t, 2H), 2.01 (d, [2H),] 1.57 (m, 2H), 1.35 (s, 12H).
References:
WO2004/13134,2004,A2 Location in patent:Page 30

38041-19-9
321 suppliers
$6.00/1g

380499-68-3
26 suppliers
$495.39/5MG

656239-37-1
23 suppliers
inquiry

180516-87-4
227 suppliers
$8.00/1g

656239-37-1
23 suppliers
inquiry