
4-[4-(2-BROMO-2-METHYL-1-OXOPROPYL)PHENYL]-CYCLOHEXANEACETIC ACID ETHYL ESTER synthesis
- Product Name:4-[4-(2-BROMO-2-METHYL-1-OXOPROPYL)PHENYL]-CYCLOHEXANEACETIC ACID ETHYL ESTER
- CAS Number:701232-18-0
- Molecular formula:C20H27BrO3
- Molecular Weight:395.33
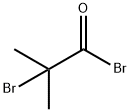
20769-85-1
257 suppliers
$13.00/5g

411238-92-1
13 suppliers
inquiry
![4-[4-(2-BROMO-2-METHYL-1-OXOPROPYL)PHENYL]-CYCLOHEXANEACETIC ACID ETHYL ESTER](/CAS/GIF/701232-18-0.gif)
701232-18-0
9 suppliers
$495.00/500mg
Yield:-
Reaction Conditions:
with aluminum (III) chloride in dichloromethane at 0;Friedel Crafts Acylation;
Steps:
1
Anhydrous AlCl3 (440 mg, 3.30 mmol) was added portionwise to a solution of Compound 3 (271 mg, 1.10 mmol) in CH2C12 (1.4 mL) at 0°C and then 2-bromoisobutyryl bromide (0.14 mL, 1.10 mmol) was added dropwise. After stirring for 1 hr at 0°C, the mixture was poured into iced water and extracted with CHC13 (5 mL). The combined organic layer was washed successively with saturated aqueous NaHCO3 solution (5 mL) and brine (5 mL), dried over MgS04 and concentrated. The residue was purified by column chromatography (hexane/AcOEt=7/1) to give Compound 4 (402 mg) as a colorless oil.
References:
WO2005/72740,2005,A2 Location in patent:Page/Page column 66-67
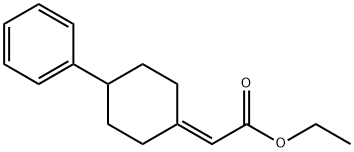
115880-04-1
23 suppliers
inquiry
![4-[4-(2-BROMO-2-METHYL-1-OXOPROPYL)PHENYL]-CYCLOHEXANEACETIC ACID ETHYL ESTER](/CAS/GIF/701232-18-0.gif)
701232-18-0
9 suppliers
$495.00/500mg
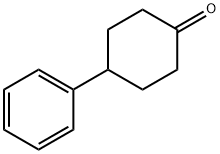
4894-75-1
168 suppliers
$6.00/5g
![4-[4-(2-BROMO-2-METHYL-1-OXOPROPYL)PHENYL]-CYCLOHEXANEACETIC ACID ETHYL ESTER](/CAS/GIF/701232-18-0.gif)
701232-18-0
9 suppliers
$495.00/500mg