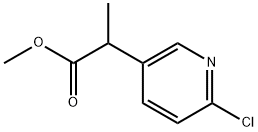
3-Pyridineacetic acid, 6-chloro-α-methyl-, methyl ester synthesis
- Product Name:3-Pyridineacetic acid, 6-chloro-α-methyl-, methyl ester
- CAS Number:717106-72-4
- Molecular formula:C9H10ClNO2
- Molecular Weight:199.63

717106-69-9
64 suppliers
$10.00/250mg

74-88-4
341 suppliers
$15.00/10g

717106-72-4
10 suppliers
inquiry
Yield:717106-72-4 1.1 g
Reaction Conditions:
Stage #1: methyl 2-(6-chloropyridin-3-yl)acetatewith n-butyllithium in tetrahydrofuran at -78; for 0.5 h;Inert atmosphere;
Stage #2: methyl iodide in tetrahydrofuran; for 12 h;Inert atmosphere;
Steps:
2.1 Step 1: Methyl 2-(6-chloropyridin-3-yl)propionate (2a)
Compound 1a (2.04g, 10.78mmol) was dissolved in THF (30mL) and cooled to -78°C,Under the protection of nitrogen, 4.74 mL of 2.5Mn-BuLi THF solution was slowly added and stirred for 30 min, and then MeI (15.61 g, 107.75 mmol) was added, and then reacted for 12 h.After the reaction was completed, the temperature was raised to room temperature, 100 mL of saturated NH4Cl solution was added to the reaction solution, and the mixture was extracted with EA (100 mL x 3).The organic phases were combined and washed with saturated brine, dried over anhydrous sodium sulfate, and then separated and purified by silica gel column chromatography (PE:EA=10:1-1:1) to obtain compound 2a (1.1 g).
References:
CN111484479,2020,A Location in patent:Paragraph 0434-0438