
Triphenyl[(tetrahydro-2H-pyran-4-yl)methyl]phosphonium iodide synthesis
- Product Name:Triphenyl[(tetrahydro-2H-pyran-4-yl)methyl]phosphonium iodide
- CAS Number:745052-92-0
- Molecular formula:C24H26IOP
- Molecular Weight:488.35
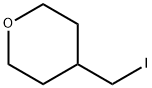
101691-94-5
113 suppliers
$22.00/0.25/ G
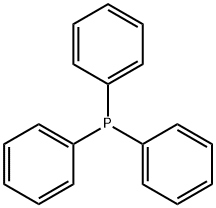
603-35-0
698 suppliers
$5.00/5G
![Triphenyl[(tetrahydro-2H-pyran-4-yl)methyl]phosphonium iodide](/CAS2/GIF/745052-92-0.gif)
745052-92-0
20 suppliers
inquiry
Yield:745052-92-0 80%
Reaction Conditions:
in acetonitrile; for 27 h;Heating / reflux;
Steps:
8
Preparation 8: Triphenyl(tetrahydropyran4-ylmethyl)phosphonium iodide; PPh,iA mixture of Preparation 7 (350g5 1.55M) and triphenylphosphine (406g, 1.55M) in acetonitrile (1.6L) was heated under reflux. After 27h the mixture was cooled and filtered, washed with diethyl ether and dried in air to provide a white solid (504g). Filtrate and washings were returned to reflux and concentrated to 750mL, reflux was maintained for 16h before cooling and dilution with diethyl ether (ca 1,2L). A precipitate formed which was stirred for 30min before being filtered, washed with diethyl ether (2 x 300mL) and dried in air to yield a further crop (lOOg). Overall yield of the title compound (604 g, 80%). RT = 2.7min; m/z (ES+) = 361.2.
References:
WO2006/16178,2006,A1 Location in patent:Page/Page column 12
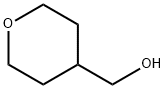
14774-37-9
246 suppliers
$5.00/1g
![Triphenyl[(tetrahydro-2H-pyran-4-yl)methyl]phosphonium iodide](/CAS2/GIF/745052-92-0.gif)
745052-92-0
20 suppliers
inquiry
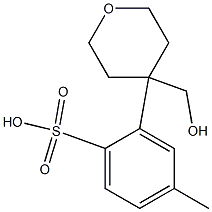
101691-65-0
52 suppliers
$17.00/250mg
![Triphenyl[(tetrahydro-2H-pyran-4-yl)methyl]phosphonium iodide](/CAS2/GIF/745052-92-0.gif)
745052-92-0
20 suppliers
inquiry