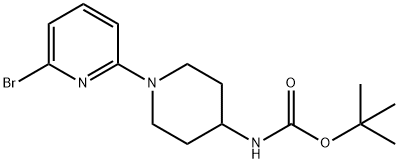
(6'-BroMo-3,4,5,6-tetrahydro-2H-[1,2']bipyridinyl-4-yl)-carbaMic acid tert-butyl ester, 98+% C15H22BrN3O2, MW: 356.27 synthesis
- Product Name:(6'-BroMo-3,4,5,6-tetrahydro-2H-[1,2']bipyridinyl-4-yl)-carbaMic acid tert-butyl ester, 98+% C15H22BrN3O2, MW: 356.27
- CAS Number:848500-12-9
- Molecular formula:C15H22BrN3O2
- Molecular Weight:356.26
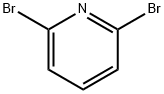
626-05-1
498 suppliers
$9.00/10g
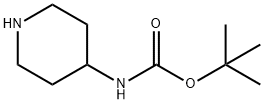
73874-95-0
456 suppliers
$10.00/250mg
![(6'-BroMo-3,4,5,6-tetrahydro-2H-[1,2']bipyridinyl-4-yl)-carbaMic acid tert-butyl ester, 98+% C15H22BrN3O2, MW: 356.27](/CAS/GIF/848500-12-9.gif)
848500-12-9
27 suppliers
$195.00/250mg
Yield:848500-12-9 32%
Reaction Conditions:
at 130 - 160; for 9.5 h;
Steps:
64.A
A mixture of 2,6-dibromo-pyridine (3.5 g, 14.77 mmol) and piperidin-4-yl-carbamic acid te/f-butyl ester (1.478 g, 7.38 mmol) is heated to 130 0C in a sealed vessel for 6.5 h and 160 0C for 3 h. The residue is cooled and dissolved in CH2CI2 , washed with saturated NaHCO3 (X2), brine, dried (Na2SO4), filtered and concentrated. The residue is then separated via flash chromatography (SiO2, 10-30% EtOAc/hexanes gradient) to give an intermediate, (6'-bromo-3,4,5,6-tetrahydro-2H-[1 ,2']bipyridinyl-4-yl)-carbamic acid te/f-butyl ester (0.83 g, 32%). MS (ESI) m/z 356.0, 358.0 (M+1 ), which is taken on to next step.
References:
WO2009/150230,2009,A1 Location in patent:Page/Page column 179