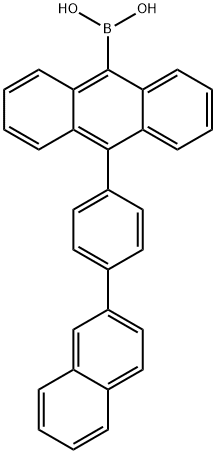
10-[4-(2-naphthalenyl)phenyl]-9-anthracenyl]-Boronic acid synthesis
- Product Name:10-[4-(2-naphthalenyl)phenyl]-9-anthracenyl]-Boronic acid
- CAS Number:853945-48-9
- Molecular formula:C30H21BO2
- Molecular Weight:424.3
![9-broMo-10-[4-(2-naphthalenyl)phenyl]Anthracene](/CAS/GIF/866611-29-2.gif)
866611-29-2
52 suppliers
$110.00/200mg
![10-[4-(2-naphthalenyl)phenyl]-9-anthracenyl]-Boronic acid](/CAS/20180808/GIF/853945-48-9.gif)
853945-48-9
14 suppliers
inquiry
Yield:853945-48-9 67%
Reaction Conditions:
Stage #1: 9-bromo-10-(4-(naphthalen-2-yl)phenyl)anthracenewith n-butyllithium in diethyl ether;hexane;toluene at -40 - -10; for 0.5 h;
Stage #2: with Trimethyl borate in diethyl ether;hexane;toluene at -70 - 20;
Stage #3: with hydrogenchloride in diethyl ether;hexane;water;toluene;
Steps:
5
9-Bromo-10-(4-naphthalene-2-yl-phenyl)anthracene 13.8 g was dispersed in dehydrated toluene 80 ml and dehydrated ether 80 ml, and the mixture was cooled down to -30°C. A 1.58M n-butyllithium hexane solution 21. 0 ml was dropwise added thereto, and after the solution was stirred at -40°C for 30 minutes, the temperature was elevated up to -10°C. The solution was cooled again down to - 70°C, and a dehydrated ether solution of trimethyl borate 10.0 ml was gradually added thereto. The solution was stirred at -70°C for 2 hours and then gradually heated up to room temperature. After left standing for a night, a 10 % hydrochloric acid aqueous solution 100 ml was added thereto and stirred, and then it was extracted twice with toluene. The organic layer was washed with saturated brine and dried on anhydrous sodium sulfate. The solvent was distilled off, and the residue was crystallized from toluene/hexane, filtered and dried, whereby targeted 10-(4-naphthalene-2-yl-phenyl)anthracene-9-boronic acid 8.48 g was obtained in the form of yellow crystal (yield: 67 %).
References:
EP1707550,2006,A1 Location in patent:Page/Page column 34