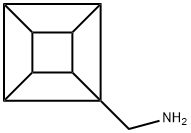
cuban-1-ylmethanamine synthesis
- Product Name:cuban-1-ylmethanamine
- CAS Number:150234-60-9
- Molecular formula:C9H11N
- Molecular Weight:133.19
![Pentacyclo[4.2.0.02,5.03,8.04,7]octanecarboxamide (9CI)](/CAS/GIF/119696-06-9.gif)
119696-06-9
3 suppliers
inquiry

150234-60-9
7 suppliers
inquiry
Yield:150234-60-9 77%
Reaction Conditions:
with lithium aluminium tetrahydride in tetrahydrofuran at 0; for 16 h;Reflux;
Steps:
pentacyclo[4.2.0.02,5.03,8.04,7]octane-1-methanamine (22)
Solid 21 (633 mg, 4.27 mmol) was dissolved in oxalyl chloride (6.41 mL) and stirred for 1 h. Excess oxalyl chloride was removed under reduced pressure and the obtained residue was dissolved in CH2Cl2 (20 mL) in a flask fitted with a cold finger containing dry-ice/acetone. Ammonia was condensed into the flask (ca. 10 mL) and the mixture allowed to stir for 1 h. The cold finger was removed and the excess ammonia and CH2Cl2 evaporated under a stream of nitrogen gas. The residue was treated with H2O (50 mL) and the aqueous phase extracted with chloroform (3 × 50 mL). The combined organic layers were dried (Na2SO4), the solvent evaporated under reduced pressure, and the obtained amide dried in vacuo. The intermediate amide was then added portionwise to a cooled (0 °C) suspension of lithium aluminium hydride (648 mg, 17.1 mmol, 4.0 equiv.) in THF (80 mL). The mixture was heated at reflux for 16 h, cooled to 0 °C, and quenched by the addition of H2O (0.65 mL), 15% aqueous sodium hydroxide (0.65 mL), and H2O (1.95 mL). The mixture was filtered, the filtrate evaporated, and the residue subjected to flash chromatography on silica gel, eluting with CHCl3-MeOH-NH4OH (90:9:1), to furnish 22 as a pale yellow oil (440 mg, 77% over two steps). The spectroscopic data for 22 matched those previously reported: δH (200 MHz; CDCl3) 4.09-3.99 (1 H, m), 3.92-3.77 (6 H, m), 2.83 (2 H, s), 1.25 (2 H, br s).
References:
Beinat, Corinne;Banister, Samuel D.;Hoban, Jane;Tsanaktsidis, John;Metaxas, Athanasios;Windhorst, Albert D.;Kassiou, Michael [Bioorganic and Medicinal Chemistry Letters,2014,vol. 24,# 3,p. 828 - 830] Location in patent:supporting information
![Pentacyclo[4.2.0.02,5.03,8.04,7]octanecarboxylic acid (7CI,9CI)](/CAS/GIF/53578-15-7.gif)
53578-15-7
31 suppliers
$459.25/5MG

150234-60-9
7 suppliers
inquiry
![Pentacyclo[4.2.0.02,5.03,8.04,7]octanecarbonyl chloride (9CI)](/CAS/GIF/60462-14-8.gif)
60462-14-8
2 suppliers
inquiry

150234-60-9
7 suppliers
inquiry
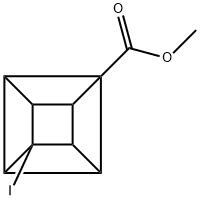
122200-58-2
11 suppliers
inquiry

150234-60-9
7 suppliers
inquiry
![Pentacyclo[4.2.0.02,5.03,8.04,7]octane-1-carboxylic acid, methyl ester](/CAS/20200611/GIF/29412-63-3.gif)
29412-63-3
3 suppliers
inquiry

150234-60-9
7 suppliers
inquiry