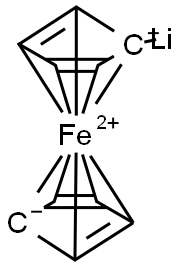
Lithium, ferrocenyl-(8CI,9CI) synthesis
- Product Name:Lithium, ferrocenyl-(8CI,9CI)
- CAS Number:1271-15-4
- Molecular formula:C10FeLi
- Molecular Weight:182.9
Yield:1271-15-4 59%
Reaction Conditions:
in tetrahydrofuran;hexane at -78 - 0;Inert atmosphere;
Steps:
General procedures
tBuLi (45 mL of a 1.6 M solution in hexanes, 72 mmol) was added over 15 min to a suspension of ferrocene (16.0 g, 86 mmol) in THF (80 mL) at 0 °C.
Immediately after the addition of t-BuLi, hexane (150 mL) was added and the suspension cooled to -78 °C.
The resulting pyrophoric orange precipitate was separated by filtration at -78 °C, washed with pre-cooled hexane (4 * 50 mL) at -78 °C and volatiles removed in vacuo to afford an orange powder (approximately 15 g).
This powder was continuously extracted with hexane (200 mL) using a Soxhlet apparatus over a period of 12 h, until the extracts were colourless.
The remaining solid was dried in vacuo to afford the target material as a highly pyrophoric orange powder.
Yield 8.17 g, 59%.
References:
Kelly, Michael J.;Tirfoin, Remi;Gilbert, Jessica;Aldridge, Simon [Journal of Organometallic Chemistry,2014,vol. 769,p. 11 - 16]
1273-73-0
96 suppliers
$38.50/500mg

1271-15-4
1 suppliers
inquiry
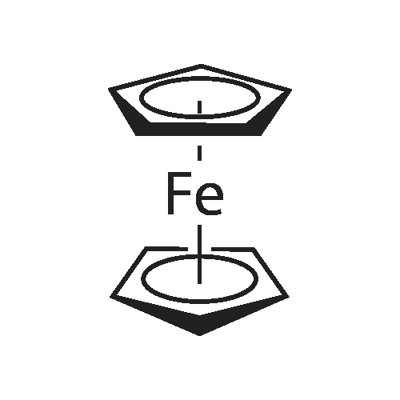
102-54-5
450 suppliers
$5.00/25g

1271-15-4
1 suppliers
inquiry
1273-76-3
23 suppliers
inquiry

1271-15-4
1 suppliers
inquiry