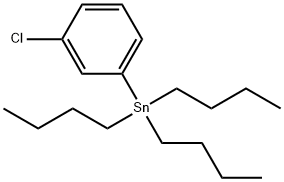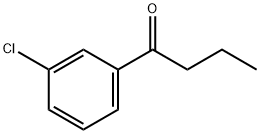
m-Chlorobutyrophenone synthesis
- Product Name:m-Chlorobutyrophenone
- CAS Number:21550-08-3
- Molecular formula:C10H11ClO
- Molecular Weight:182.65
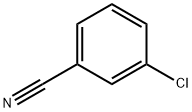
766-84-7
256 suppliers
$9.00/10g
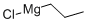
2234-82-4
99 suppliers
$67.50/100ml

21550-08-3
17 suppliers
$183.33/1g
Yield:21550-08-3 97%
Reaction Conditions:
Stage #1: 3-chloro-benzonitrile;1-propylmagnesium chloride;copper(l) chloride in tetrahydrofuran;diethyl ether; for 0.5 h;Heating / reflux;
Stage #2: with hydrogenchloride in tetrahydrofuran;diethyl ether;water at 20;
Stage #3: with water;hydrogenchloride at 90; for 1 h;
Steps:
5A.a
Preparation of N-(2-butyryl-4-chlorophenyl)trifluoromethanesulfonamide (Formula 29A) a) To a stirred solution of 3-chlorobenzonitrile (2.0g, 14.54 mmol) and n-propylmagnesium chloride (2M in Et2O) (8.0 mL, 15.99 mmol) in THF (40 mL) was added CuCl (29 mg, 0.29 mmol), and the mixture was refluxed for 30 minutes. After cooling to RT, cold 1N HCl (10 mL) was added cautiously, the THF removed under reduced pressure, further 1N HCl (30 mL) added and the reaction heated at 90° C. for 1 hour. To the cooled reaction mixture was added water (20 mL) and CH2Cl2 (50 mL), the phases separated, and the aqueous phase again extracted with CH2Cl2. The combined organics were washed with water, dried over MgSO4 and the solvent evaporated under vacuum. The residue was filtered through a pad of silica (eluting with CH2Cl2) to afford 1-(3-chlorophenyl)butan-1-one 26A (3.14 g, 97%), as a yellow liquid.
References:
US2006/63841,2006,A1 Location in patent:Page/Page column 33; 8/12
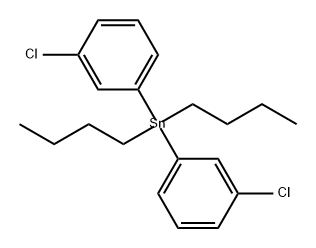
1422446-29-4
0 suppliers
inquiry

141-75-3
333 suppliers
$12.32/25G

21550-08-3
17 suppliers
$183.33/1g

683-18-1
153 suppliers
$18.64/25G
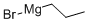
927-77-5
108 suppliers
$209.00/250g
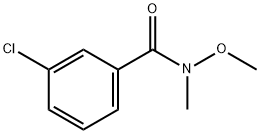
145959-21-3
69 suppliers
$52.50/250mg

21550-08-3
17 suppliers
$183.33/1g
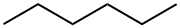
110-54-3
778 suppliers
$20.00/100ml
10557-57-0
0 suppliers
inquiry

145959-21-3
69 suppliers
$52.50/250mg

21550-08-3
17 suppliers
$183.33/1g