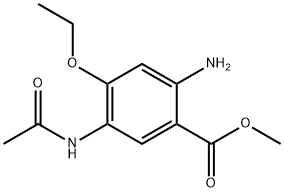
Methyl 5-acetaMido-2-aMino-4-ethoxybenzoate synthesis
- Product Name:Methyl 5-acetaMido-2-aMino-4-ethoxybenzoate
- CAS Number:1222172-49-7
- Molecular formula:C12H16N2O4
- Molecular Weight:252.27

1201080-23-0
5 suppliers
inquiry

1222172-49-7
11 suppliers
inquiry
Yield:1222172-49-7 91%
Reaction Conditions:
with hydrogen;Raney-Ni in tetrahydrofuran;methanol; for 3 h;
Steps:
3
Example 3 Methyl 4-ethoxy-5-acetylamido-2-aminobenzoate (a compound of formula II, R=methyl) The title compound of example 1 of methyl 4-ethoxy-5-acetylamido-2-nitrobenzoate (320 g, 1.13 mol) and Raney-Ni (40 g) were mixed with THF (1500 mL) and methanol (1500 mL) and stirred for 3 h under hydrogen. The reaction mixture was filtered, washed with dichloromethane, and the filtrate was distilled off the solvent to give 260 g of methyl 4-ethoxy-5-acetylamido-2-aminobenzoate with a yield of 91%. 1HNMR (300 MHz, DMSO-d6) δ 1.45 (t, 3H), 2.16 (s, 3H), 3.82 (s, 3H), 4.06 (q, 2H), 5.71 (br, 1H), 6.04 (s, 1H), 7.26 (s, 1H), 7.35 (br, 1H), 8.68 (s, 1H). MS/ESI: 252.9 (M+H).
References:
US2011/263860,2011,A1 Location in patent:Page/Page column 5

1201080-20-7
11 suppliers
inquiry

1222172-49-7
11 suppliers
inquiry
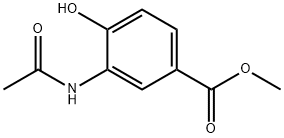
126360-59-6
0 suppliers
inquiry

1222172-49-7
11 suppliers
inquiry
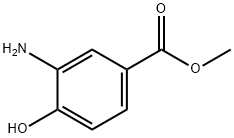
536-25-4
189 suppliers
$11.00/250mg

1222172-49-7
11 suppliers
inquiry