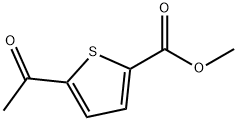
Methyl 5-Acetylthiophene-2-carboxylate synthesis
- Product Name:Methyl 5-Acetylthiophene-2-carboxylate
- CAS Number:4101-81-9
- Molecular formula:C8H8O3S
- Molecular Weight:184.21
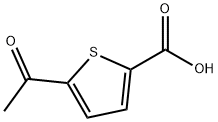
4066-41-5
210 suppliers
$6.00/1g
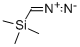
18107-18-1
210 suppliers
$33.00/5g

4101-81-9
33 suppliers
$129.00/1g
Yield:4101-81-9 100%
Reaction Conditions:
in methanol;toluene at 0 - 20;
Steps:
20.1
Step 1: methyl 5-acetylthiophene-2-carboxylateTo a solution of 5-acetylthiophene-2-carboxylic acid (3.24 g, 19.0 mmol) dissolved in a mixture of toluene (50 mL) and methanol (50 mL) cooled to 0° C. was added 2.0 M trimethylsilyldiazomethane in ether (19.0 mL, 38.1 mmol) dropwise. Upon complete addition, the reaction mixture was allowed to warm to room temperature overnight. The reaction was quenched with the addition of acetic acid and the solvent was removed. The residue thus obtained was partitioned between ethyl acetate and saturated aqueous NaHCO3 and upon separation, the organic layer was dried over anhydrous magnesium sulfate and concentrated to afford the desired product (3.49 g, 100%). LC-MS: (FA) ES+ 185)
References:
US2011/213003,2011,A1 Location in patent:Page/Page column 64
88-15-3
600 suppliers
$5.00/10g

67-56-1
738 suppliers
$9.00/25ml
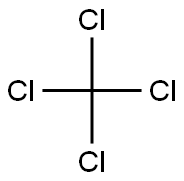
56-23-5
6 suppliers
$25.00/10ml

4101-81-9
33 suppliers
$129.00/1g
