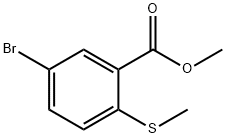
Methyl 5-bromo-2-methylthiobenzoate synthesis
- Product Name:Methyl 5-bromo-2-methylthiobenzoate
- CAS Number:929000-14-6
- Molecular formula:C9H9BrO2S
- Molecular Weight:261.14
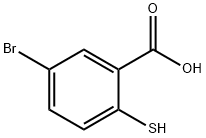
61954-80-1
34 suppliers
$184.37/100mg

74-88-4
342 suppliers
$15.00/10g

929000-14-6
24 suppliers
$110.00/1g
Yield:929000-14-6 89.3%
Reaction Conditions:
with potassium carbonate at 80; for 4 h;
Steps:
2.2 Step 2: Methyl 5-bromo-2-(methylthio)benzoate (Intermediate 203)
Potassium carbonate (83.0 g, 601 mmol, 7.0 eq.) and ethyl iodide (21.4 mL, 343 mmol, 4.0 eq.) were added 0° C. to a stirred solution of 5-bromo-2-mercaptobenzoic acid (202) (20 g, 85.8 mmol, 1 eq.) in DMF (600 mL) at RT.
The reaction mixture was stirred at 80° C. for 4 h.
The progress of the reaction was monitored by TLC (10% EtOAc in n-hexane) to ensure the completion of the reaction. DMW (500 mL) was added and the product was extracted with ethyl acetate (3*500 mL).
The combined EtOAc extract was washed with brine (300 mL), dried over sodium sulfate, filtered and filtrate was concentrated under vacuum to get the crude compound.
The crude product was purified by column chromatography on silica gel (230-400 mesh size) and gradient solvent (0-5% EtOAc in n-hexane) to afford intermediate 203 (20 g, 89.3%) as an off-white solid.
References:
US2021/101911,2021,A1 Location in patent:Paragraph 0142; 0176-0177; 0180-0181
5794-88-7
464 suppliers
$6.00/10g

929000-14-6
24 suppliers
$110.00/1g