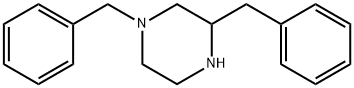
N-1-BENZYL-3-BENZYL-PIPERAZINE synthesis
- Product Name:N-1-BENZYL-3-BENZYL-PIPERAZINE
- CAS Number:179051-52-6
- Molecular formula:C18H22N2
- Molecular Weight:266.38
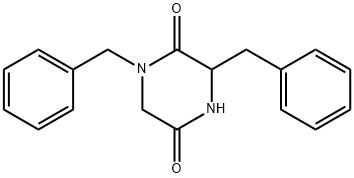
109559-14-0
5 suppliers
inquiry

179051-52-6
22 suppliers
inquiry
Yield:179051-52-6 100%
Reaction Conditions:
Stage #1: 1-benzyl-3-(R)-benzyl piperazine-2,5-dionewith lithium aluminium tetrahydride in tetrahydrofuran at 0; for 21.75 h;Heating / reflux;
Stage #2: with sodium hydroxide;water in tetrahydrofuran;
Steps:
II
(+/-)-l,3-Dibenzylpiperazine, 7.; [0027] To a 0 0C suspension of dione 6 (1.38 g, 4.7 mmol) in dry THF (120 mL) was added LiAlH4 (0.713 g, 18.8 mmol) in two portions. The mixture was maintained on ice for 15 min, then the reaction was heated at reflux for 2.5 h. The solution was cooled and stirred for an additional 19 h then the excess LiAlH4 was quenched by the careful, sequential addition of 0.7 mL water, 1.3 mL 4 M NaOH and 0.7 mL water. The solids that formed will filtered (Celite) and the filtrate concentrated to provide 7 as a pale colored solid (1.29 g, 103 %, with THF) that slowly formed from an oil. 1H NMR (400 MHz, CDCl3): δ 1.93 (m, triplet shape, IH), 2.11 (td, J= 2.9, 11.0 Hz, IH, overlapped with bs, NH, 1η), 2.58 (dd, 8.8, 13.2 Hz, IH), 2.70 - 2.77 (m, 2H), 2.79 - 2.87 (m, 2H), 2.92 (dt, J= 2.3, 11.7 Hz, IH), 3.02 (m, IH), 3.51 (dd, AB pattern, J = 12.8 Hz, 2H), 7.18 - 7.33 (m, 10H); 13C NMR (100 MHz, CDCl3): δ 40.7, 45.6, 53.3, 56.2, 59.6, 63.3, 126.3, 127.0, 128.1, 128.4, 129.2, 137.9, 138.5. HRMS Calcd. for [C18H22N2 + H+]: 267.1861. Found 267.1869.
References:
WO2008/115593,2008,A1 Location in patent:Page/Page column 13
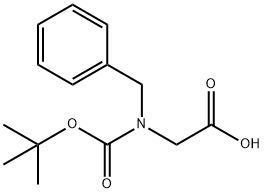
76315-01-0
53 suppliers
$82.50/1g

179051-52-6
22 suppliers
inquiry
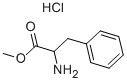
5619-07-8
108 suppliers
$6.00/5g

179051-52-6
22 suppliers
inquiry