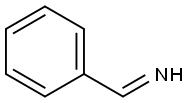
N-Benzylideneamine synthesis
- Product Name:N-Benzylideneamine
- CAS Number:16118-22-2
- Molecular formula:C7H7N
- Molecular Weight:105.14
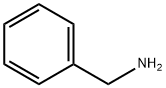
100-46-9
486 suppliers
$5.00/5 g

16118-22-2
3 suppliers
inquiry
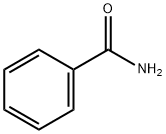
55-21-0
396 suppliers
$5.00/5 g
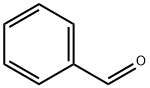
100-52-7
956 suppliers
$17.30/2g
Yield: 78.3% , 19% , 1.7%
Reaction Conditions:
with ammonia in 1,4-dioxane;water at 150; under 4500.45 Torr; for 8 h;Catalytic behavior;Reagent/catalyst;
Steps:
9; 14 Example 9; Example 14
This example describes the preparation of benzamide from benzyl amine over the non-precious metal-containing silica catalysts reported in examples 1-8. In a typical reaction, 5 mmol of benzyl amine, 15 mL of 1,4-dioxane and 1 mL of 25% ammonia solution were charged into a stainless-steel pressure reactor. 0.2 g of catalyst was added to it. The reactor was pressurized to 6 bar with air. Temperature of the reactor was raised to 150° C. and the reaction was conducted for 8 hrs while stirring at a speed of 600 revolutions per min. Then, temperature was lowered down to 25° C. and the reactor was depressurized. Catalyst was separated by centrifugation/filtration. Solvent was evaporated and the liquid portion was analyzed by gas chromatography (Varian 3400). Identity of the products was confirmed by comparing with the standard samples. Catalytic activity data of different metal-containing catalysts in the preparation of amides are listed in Table 1.; This example describes the stability and reusability of Mn-SBA-16 (Si/Mn=30) catalyst in the preparation of benzamide from benzyl amine.
The catalyst recovered after the catalytic run in Example 9 is washed with methanol, dried at 90° for 4 h and then reused in this experiment. The reaction was conducted in the same manner as described in Example 9 but with the used Mn-SBA-16 (Si/Mn=30) catalyst. This reusability experiment was carried out for three times. 1st Reuse: Benzylamine conversion=100 mol %, benzamide selectivity=78.3 mol %. imine selectivity=19.0 mol % and benzaldehyde=1.7 mol %. 2nd Reuse: Benzylamine conversion=100 mol %, benzamide selectivity=77.8 mol %. imine selectivity=19.2 mol % and benzaldehyde=2.0 mol%. 3rd Reuse: Benzylamine conversion=100 mol %, benzamide selectivity=78.0 mol %. imine selectivity=19.1 mol % and benzaldehyde=1.9 mol %.
References:
COUNCIL OF SCIENTIFIC & INDUSTRIAL RESEARCH;Srinivas, Darbha;Kumar, Anuj;Devadutta, Nepak US2015/99900, 2015, A1 Location in patent:Paragraph 0050; 0055

100-52-7
956 suppliers
$17.30/2g

16118-22-2
3 suppliers
inquiry
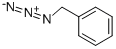
622-79-7
114 suppliers
$15.00/250mg

16118-22-2
3 suppliers
inquiry

100-46-9
486 suppliers
$5.00/5 g

16118-22-2
3 suppliers
inquiry