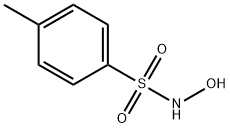
N-Hydroxy-4-methylbenzenesulfonamide synthesis
- Product Name:N-Hydroxy-4-methylbenzenesulfonamide
- CAS Number:1593-60-8
- Molecular formula:C7H9NO3S
- Molecular Weight:187.22
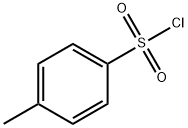
98-59-9
586 suppliers
$9.00/5g

1593-60-8
38 suppliers
$45.00/250mg
Yield: 87.5%
Reaction Conditions:
with hydroxylamine hydrochloride;magnesium oxide in tetrahydrofuran;methanol;water at 20;
Steps:
3.1 first step
Hydroxylamine hydrochloride (7.2 g, 86 mmol) was dissolved in methanol (30 ml) and water (20 ml), magnesium oxide (5.1 g, 129 mmol) was added, stirred for 10 minutes, then p-toluenesulfonyl chloride (8 g, 43 mmol) in tetrahydrofuran (300 ml), stirred vigorously at room temperature, and reacted to TLC to monitor the starting material for complete reaction. The extract was filtered through Celite, dried over anhydrous magnesium sulfate and concentrated under reduced pressure to give N-hydroxy-4-methylbenzenesulfonamide (7 g, white solid) in 87.5% yield.
References:
Shanghai Acebright Pharmaceuticals Group Co., Ltd.;An, Xiaoxia;Bie, Pingyan;Liu, Jun;Zhuang, Geshi CN105418632, 2016, A Location in patent:Paragraph 0175; 0176; 0177

70-55-3
569 suppliers
$6.00/100g

1593-60-8
38 suppliers
$45.00/250mg
66021-66-7
0 suppliers
inquiry

1593-60-8
38 suppliers
$45.00/250mg