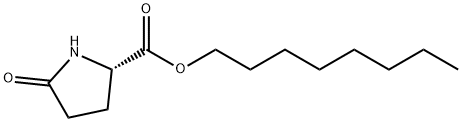
N-OCTYL L-2-PYRROLIDONE-5-CARBOXYLATE synthesis
- Product Name:N-OCTYL L-2-PYRROLIDONE-5-CARBOXYLATE
- CAS Number:4931-70-8
- Molecular formula:C13H23NO3
- Molecular Weight:241.33

111-87-5
723 suppliers
$6.00/100g
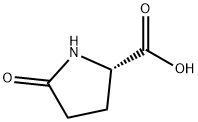
149-87-1
311 suppliers
$6.00/25g
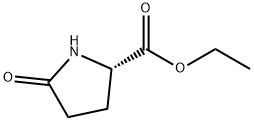
66183-71-9
41 suppliers
$17.00/5g

4931-70-8
18 suppliers
$47.70/5g
Yield:4931-70-8 100%
Reaction Conditions:
Novozym 435 at 60; under 15.0015 Torr; for 24 h;Enzymatic reaction;
Steps:
4 Preparation of Octyl Pyroglutamate
In a multinecked round-bottomed flask, 50.0 g (336 mmol) of a mixture of ethyl pyroglutamate and pyroglutamic acid (molar ratio 71:29) and 43.7 g (336 mmol) of octanol as initial charge were heated to 60° C. After adding 4.6 g of Novozym 435 (immobilized lipase B from C. antarctica), a vacuum was applied (20 mbar) and low-boiling reaction products were distilled off. The reaction was monitored by means of NMR spectroscopy or by means of a suitable chromatographic method. Conversion after 8 hours: 95%, after 24 hours: >98%. When the reaction was complete, the immobilized enzyme was filtered off. The filtrate gave 81 g of product (100% of the theoretical yield) without further work-up as a pale yellow liquid. 1H NMR (DMSO-d6, 400 MHz): δ=8.0 (br, s, 1H), 4.2 (dd, 3J=8.6 Hz, 4.3 Hz, 1H), 4.1 (t, 3J=6.5 Hz, 2H), 2.3 (m, 1H), 2.1 (m, 2H), 2.0 (m, 1H), 1.6 (m, 2H), 1.3 (m, 10H), 0.8 (t, 3J=7.1 Hz, 3H).
References:
US2006/74259,2006,A1 Location in patent:Page/Page column 3