
tert-Butyl 4-(chloromethyl)thiazol-2-ylcarbamate synthesis
- Product Name:tert-Butyl 4-(chloromethyl)thiazol-2-ylcarbamate
- CAS Number:892952-70-4
- Molecular formula:C9H13ClN2O2S
- Molecular Weight:248.73
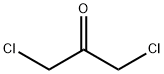
534-07-6
9 suppliers
$10.00/1g
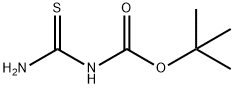
268551-65-1
47 suppliers
$29.19/250mg

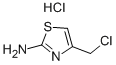
892952-70-4
33 suppliers
$137.75/500mg
Yield:892952-70-4 68%
Reaction Conditions:
Stage #1: 1,3-Dichloroacetone;tert-butyl carbamothioylcarbamate in acetone at 20; for 72 h;
Stage #2: with water;sodium hydrogencarbonate in acetone; for 0.25 h;Product distribution / selectivity;
Steps:
9
Example 9: Preparation of /V-Boc-4-(chIoromethyl)thiazol-2-amine (Intermediate VIII); [0095] To a solution of Intermediate VII (3.0 g, 17 mmol) in acetone (75 mL) was added 1,3-dichloroacetone (2.4 g, 18.7 mmol) at once. The mixture was stirred at room temperature for 72 h. The reaction mixture was treated with NaHCCb (3 g) and stirred for 15 min. The solids were removed by filtration and the filtrate was concentrated in vacuo. Column chromatography (EtOAc/Heptane, 4/1) afforded N-Boc-4-(chloromethyl)thiazol-2- amine (2.9 g, 1 1.6 mmol, 68%) as a white solid.
References:
WO2008/11191,2008,A1 Location in patent:Page/Page column 38

24424-99-5
824 suppliers
$13.50/25G
59608-97-8
115 suppliers
$17.00/500mg

892952-70-4
33 suppliers
$137.75/500mg