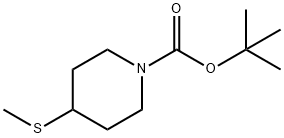
tert-butyl 4-(methylthio)piperidine-1-carboxylate synthesis
- Product Name:tert-butyl 4-(methylthio)piperidine-1-carboxylate
- CAS Number:208245-69-6
- Molecular formula:C11H21NO2S
- Molecular Weight:231.35
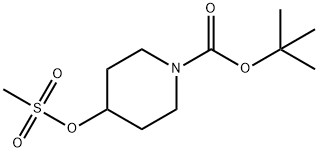
141699-59-4
233 suppliers
$6.00/1g
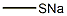
5188-07-8
265 suppliers
$13.00/5g

208245-69-6
21 suppliers
inquiry
Yield:208245-69-6 67%
Reaction Conditions:
with tetra-(n-butyl)ammonium iodide in tetrahydrofuran at 20; for 72 h;
Steps:
60 Reference Example 60
A mixture of tert-butyl 4-(methylsulfonyl)piperidine-1-carboxylate (12.5 g), methylmercaptan sodium salt (3.31 g), tetra-n-butylammonium iodide (1.66 g) and tetrahydrofuran (50 mL) was stirred at room temperature for 72 hrs. The insoluble material was filtered through celite and the filtrate was concentrated. The residue was subjected to silica gel column chromatography, and tert-butyl 4-(methylthio)piperidine-1-carboxylate was obtained as a colorless oil from a fraction eluted with ethyl acetate-hexane (1:4, volume ratio) (6.89 g, yield 67%). NMR(CDCl3) delta:1.45(9H, s), 1.40-1.60(2H, m), 1.82-2.00(2H, m), 2.10(3H, s), 2.60-2.80(1H, m), 2.80-3.00(2H, m), 3.92-4.08(2H, m).
References:
EP1486490,2004,A1 Location in patent:Page 46

141699-59-4
233 suppliers
$6.00/1g

208245-69-6
21 suppliers
inquiry
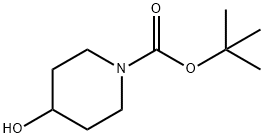
109384-19-2
470 suppliers
$5.00/1g

208245-69-6
21 suppliers
inquiry

24424-99-5
825 suppliers
$13.50/25G

208245-69-6
21 suppliers
inquiry