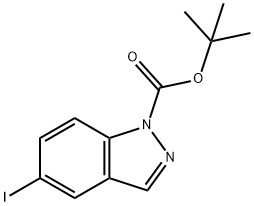
tert-butyl 5-iodo-1H-indazole-1-carboxylate synthesis
- Product Name:tert-butyl 5-iodo-1H-indazole-1-carboxylate
- CAS Number:1001907-23-8
- Molecular formula:C12H13IN2O2
- Molecular Weight:344.15

24424-99-5
825 suppliers
$13.50/25G
55919-82-9
148 suppliers
$6.00/1g

1001907-23-8
22 suppliers
inquiry
Yield:-
Reaction Conditions:
with 2-(Dimethylamino)pyridine;triethylamine in acetonitrile at 0 - 20; for 3 h;
Steps:
18.A 5-(4-fluorophenylsulfanyl)-1-H-indazole
Step A: A mixture of 6-iodo-1H-indazole (compound 1d) in CH3CN (11 mL) was treated with triethylamine and dimethylaminopyridine. After cooling to 0 C., a solution of di-tert-butyl dicarbonate (BOC anhydride) in CH3CN (10 mL) was added dropwise. After stirring at room temperature for 3 hours, the reaction mixture was concentrated in vacuum and the resulting residue was partitioned between H2O and ether. The pH was adjusted to 2 with 1N HCl and the organic phase was separated, dried (Na2SO4), filtered and concentrated in vacuum to provide compound 2d as an oil.
References:
Munson, Mark;Rizzi, James;Rodriguez, Martha;Kim, Ganghyeok US2004/176325, 2004, A1 Location in patent:Page/Page column 24; sheet 20

850363-46-1
2 suppliers
inquiry

1001907-23-8
22 suppliers
inquiry
13194-68-8
206 suppliers
$6.00/1g

1001907-23-8
22 suppliers
inquiry