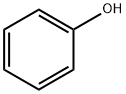
페놀
|
|
페놀 속성
- 녹는점
- 40-42 °C(lit.)
- 끓는 점
- 182 °C(lit.)
- 밀도
- 1.071 g/mL at 25 °C(lit.)
- 증기 밀도
- 3.24 (vs air)
- 증기압
- 0.09 psi ( 55 °C)
- 굴절률
- n
20/D 1.53
- FEMA
- 3223 | PHENOL
- 인화점
- 175 °F
- 저장 조건
- 2-8°C
- 용해도
- H2O: 50 mg/mL at 20 °C, 투명, 무색
- 물리적 상태
- 액체
- 산도 계수 (pKa)
- 9.89(at 20℃)
- Specific Gravity
- 1.071
- 색상
- 희미하게 노란색
- 수소이온지수(pH)
- 6.47(1 mM solution);5.99(10 mM solution);5.49(100 mM solution);
- 냄새
- 0.06ppm에서 감지 가능한 달콤한 약 냄새
- 폭발한계
- 1.3-9.5%(V)
- Odor Threshold
- 0.0056ppm
- ?? ??
- 페놀성
- 수용성
- 8g/100mL
- 어는점
- 41℃
- 감도
- Air & Light Sensitive
- Merck
- 14,7241
- JECFA Number
- 690
- BRN
- 969616
- Henry's Law Constant
- 1.09 at 5 °C (average derived from six field experiments, Lüttke and Levsen, 1997)
- Dielectric constant
- 4.3(10℃)
- 노출 한도
- TLV-TWA skin 5 ppm (~19 mg/m3 ) (ACGIH, MSHA, and OSHA); 10-hour TWA 5.2 ppm (~20 mg/m3 ) (NIOSH); ceiling 60 mg (15 minutes) (NIOSH); IDLH 250 ppm (NIOSH).
- 안정성
- 흡습성
- InChIKey
- ISWSIDIOOBJBQZ-UHFFFAOYSA-N
- LogP
- 1.47 at 30℃
- CAS 데이터베이스
- 108-95-2(CAS DataBase Reference)
- IARC
- 3 (Vol. 47, 71) 1999
- NIST
- Phenol(108-95-2)
안전
- 위험 및 안전 성명
- 위험 및 사전주의 사항 (GHS)
| 위험품 표기 | T,C,F,Xn | ||
|---|---|---|---|
| 위험 카페고리 넘버 | 23/24/25-34-48/20/21/22-68-40-39/23/24/25-11-36-20/21/22-24/25 | ||
| 안전지침서 | 26-36/37/39-45-36/37-28A-28-24/25-1/2-36-16-7 | ||
| 유엔번호(UN No.) | UN 2821 6.1/PG 2 | ||
| WGK 독일 | 2 | ||
| RTECS 번호 | SJ3325000 | ||
| F 고인화성물질 | 8-23 | ||
| 자연 발화 온도 | 715 °C | ||
| TSCA | Yes | ||
| 위험 등급 | 6.1 | ||
| 포장분류 | II | ||
| HS 번호 | 29071100 | ||
| 유해 물질 데이터 | 108-95-2(Hazardous Substances Data) | ||
| 독성 | LD50 orally in rats: 530 mg/kg (Deichmann, Witherup) | ||
| IDLA | 250 ppm | ||
| 기존화학 물질 | KE-28209 | ||
| 유해화학물질 필터링 | 97-1-332 | ||
| 사고대비 물질 필터링 | 29 | ||
| 함량 및 규제정보 | 물질구분: 유독물질; 혼합물(제품)함량정보: 페놀 및 이를 5% 이상 함유한 혼합물 |
페놀 C화학적 특성, 용도, 생산
물성
페놀은 비록 여전히 약산성으로 고려되지만 지방족 알코올에 비해 페놀은 약 10억 배 산성이 더 강하다. 페놀은 NaOH 수용액과 완전히 반응하여 H+를 일지만 대부분의 알코올은 단지 부분적으로 반응하나.용도
많은 물질들과 합성물의 전구체(precursor)로서 중요한 산업 물질이다. 주로 플라스틱 또는 플라스틱과 관련된 물질들로 변환된다. 페놀과 페놀의 화학적 파생물은 폴리카보네이트, 나일론, 세제, 제초제, 수많은 약들의 주요 성분이다.용도
페놀은 조셉 리스터에 의해 수술을 위한 살균제로 쓰였다. 구강 마취제나 독감 치료제로 쓰이기도 하였다. 페놀은 아스피린과 같은 약품의 제조 공정에 사용되며 제초제의 원료로도 사용되었다. 레오 베이클랜드는 페놀을 이용하여 최초의 플라스틱인 베이클라이트를 합성하였다. 페놀은 인쇄회로기판(PCB)의 제조 공정에 사용된다. 사용한 폐 페놀을 안전하게 처리하지 않을 경우 수질오염을 유발시킬 수 있다.독성
페놀은 소화기, 호흡, 피부 접촉등을 통해 인체에 흡수될 경우 심각한 장애나 사망에 이를 수 있는 맹독 물질이다. 경구를 통해 소화기에서 흡수되는 경우 치사량은 1 ~ 15g이다. 4.8g을 섭취하고 10분 내에 사망한 경우가 보고되어 있다.개요
Phenol is a stable chemical substance and appear as colourless/white crystals with a characteristic, distinct aromatic/acrid odour. It is reactive and incompatible with strong oxidising agents, strong bases, strong acids, alkalis, and calcium hypochlorite. Phenol is flammable and may discolour in light. Phenol is used in the manufacture or production of explosives, fertiliser, coke, illuminating gas, lampblack, paints, paint removers, rubber, perfumes, asbestos goods, wood preservatives, synthetic resins, textiles, drugs, and pharmaceutical preparations. It is also extensively used as a disinfectant in the petroleum, leather, paper, soap, toy, tanning, dye, and agricultural industries.화학적 성질
Phenol, C6H5OH, also known as carbolic acid and phenylic acid, is a white poisonous crystalline solid that melts at 43 °C (110 OF) and boils at 182°C (360 OF). Phenol has a sharp burning taste,a distinctive odor, and it irritates tissue. It is toxic not only by ingestion or inhalation, but also by skin absorption. Phenol is soluble in water,alcohol,and ether. It is used in the production of resins,germicides,weedkillers,pharmaceuticals, and as a solvent in the refining of lubricating oils.물리적 성질
Phenol is a colorless or white crystalline solid that is slightly soluble in water. Phenol is the simplest of the large group of organic chemicals known as phenols, which consist of compounds where a carbon in the phenyl aromatic group (C6H5) is directly bonded to hydroxyl, OH.출처
It is reported found in over 150 natural products including apricot, sour cherry, black currant, bilberry, cranberry, other berries, grapes, guava fruit, peach, pineapple, asparagus, onion, cooked potato, tomato, cinnamon bark, cassia leaf, ginger, pennyroyal oil, many cheeses, butter, milk, milk powder, boiled egg, fish and fish oil, cooked and cured meats, beer, wheaten bread, crisp bread, cognac, rose wine, cocoa, coffee, tea, whiskies, roasted filbert, roasted peanut, soybean, pecans, honey, avocado, Arctic bramble, passion fruit, beans, mushrooms, burley tobacco, cooked beef and chicken, fermented soy sauce, trassi, roasted almonds, sesame seed, fenugreek, mango, tamarind, Brazil nut, rice, rhubarb, licorice, buckwheat, watercress, malt, wort, dried bonito, loquat, myrtle berry, rosemary, Tahiti and Bourbon vanilla, endive, shrimp, crab, crayfish, clam, squid, truffle and Chinese quince.역사
Phenol’s first prominent use was by Joseph Lister (1827–1912) as an antiseptic. Throughout human history,infection often resulted in death,even when the wound could be surgically treated.A broken bone piercing the skin, which today is a painful but not life-threatening injury,historically resulted in infection and possible amputation or death. Lister was inspired by Louis Pasteur’s (1822–1895) germ theory of disease,and he began to use antiseptic methods during routine surgery during the 1860s.용도
phenol is frequently used for medical chemical face peels. It may trap free radicals and can act as a preservative. Phenol, however, is an extremely caustic chemical with a toxicity potential. It is considered undesirable for use in cosmetics. even at low concentrations, it frequently causes skin irritation, swelling, and rashes.정의
ChEBI: An organic hydroxy compound that consists of benzene bearing a single hydroxy substituent. The parent of the class of phenols.생산 방법
Historically, phenol was produced by the distillation of coal tar. Today, phenol is prepared by one of several synthetic methods, such as the fusion of sodium benzenesulfonate with sodium hydroxide followed by acidification; the hydrolysis of chlorobenzene by dilute sodium hydroxide at high temperature and pressure to give sodium phenate, which on acidification liberates phenol (Dow process); or the catalytic vapor-phase reaction of steam and chlorobenzene at 500°C (Raschig process).제조 방법
Phenol is formed in dry distillation of wood, peat and coal; coal tar is one of the commercial sources of phenol and its homologues.Indications
Phenol in dilute solution (0.5% to 2%) decreases itch by anesthetizing the cutaneous nerve endings. Phenol should never be used on pregnant women or infants younger than 6 months of age.World Health Organization (WHO)
Phenol became widely used as an antiseptic following demonstration of its germicidal activity in 1867. It is an intensely corrosive substance and percutaneous absorption can produce serious systemic toxicity. It has been withdrawn from pharmaceutical preparations by at least one national regulatory authority. However, it is still used widely in concentrations of the order of 1.4% in proprietary preparations for the relief of soreness of the mouth and throat.일반 설명
A solid melting at 110°F. Colorless if pure, otherwise pink or red. Flash point 175°F. Density 9.9 lb / gal. Vapors are heavier than air Corrosive to the skin (turning skin white) but because of its anesthetic quality numbs rather than burn. Lethal amounts can be absorbed through the skin. Used to make plastics and adhesives.공기와 물의 반응
Decomposes slowly in air. Mixtures of 9-10% phenol in air are explosive. Soluble in water반응 프로필
PHENOL is a weak acid. Reacts exothermically with bases. Reacts with strong oxidizing agents. Emits acrid smoke and irritating fumes when heated to decomposition. Undergoes, in the presence of aluminum chloride, potentially explosive reactions with nitromethane, butadiene, formaldehyde, peroxodisulfuric acid, peroxosulfuric acid, and sodium nitrite . Reacts violently with sodium nitrate in the presence of trifluoroacetic acid [Bretherick, 5th ed., 1995, p. 770]. May corrode lead, aluminum and its alloys, certain plastics, and rubber. Phenol may explode in contact with peroxodisulfuric acid (Dns, J. Ber., 1910, 43, 1880; Z. Anorg. Chem., 1911, 73, 1911.) or peroxomonosulfuric acid. (Sidgwick, 1950, 939)건강위험
Phenol and its vapors are corrosive to the eyes, skin, and respiratory tract. The corrosive effect on skin and mucous membranes is due to a protein-degenerating effect. Repeated or prolonged skin contact with phenol may cause dermatitis, and potentially second and third-degree burns. Inhalation of phenol vapor may cause lung edema. Phenol may adversely effect the central nervous system and heart. Long-term, or repeated exposure, to phenol may have harmful effects on the liver and kidneys.While there is no evidence that phenol causes cancer in humans it is readily absorbed through the skin; systemic poisoning can occur in addition to the local caustic burns. Resorptive poisoning by a large quantity of phenol can occur even with only a small area of skin, rapidly leading to paralysis of the central nervous system and a severe drop in body temperature. Phenol is also a reproductive toxin causing increased risk of abortion and low birth weight indicating retarded development in utero.
Chemical burns from skin exposures can be decontaminated by washing with polyethylene glycol or isopropyl alcohol; flushing with copious amounts of water will help to remediate the burn. Removal of contaminated clothing is required, as well as immediate hospital treatment for large splashes.
https://ehs.ucsc.edu/lab-safety-manual/specialty-chemicals/phenol.html
화재위험
Flammable vapors when heated. Runoff from fire control water may give off poisonous gases and cause pollution. Mixtures of 9-10% phenol in air are explosive. Avoid aluminum chloride/nitrobenzene mixture, peroxodisulfuric acid, peroxomonosulfuric acid and strong oxidizing agents. Decomposes slowly on air contact. Avoid contact with strong oxidizing agents.인화성 및 폭발성
Phenol is a combustible solid (NFPA rating = 2). When heated, phenol produces flammable vapors that are explosive at concentrations of 3 to 10% in air. Carbon dioxide or dry chemical extinguishers should be used to fight phenol fires.Pharmaceutical Applications
Phenol is used mainly as an antimicrobial preservative in parenteral pharmaceutical products. It has also been used in topical pharmaceutical formulations and cosmetics;Phenol is widely used as an antiseptic, disinfectant, and therapeutic agent, although it should not be used to preserve preparations that are to be freeze-dried.
공업 용도
Phenol is the simplest member of a class oforganic compounds possessing a hydroxylgroup attached to a benzene ring or to a morecomplex aromatic ring system.Also known as carbolic acid or monohydroxybenzene,phenol is a colorless to whitecrystalline material of sweet odor, having thecomposition C6H5OH, obtained from the distillationof coal tar and as a by-product ofcoke ovens.
Phenol has broad biocidal properties, anddilute aqueous solutions have long been usedas an antiseptic. At higher concentrations itcauses severe skin burns; it is a violent systemicpoison. It is a valuable chemical raw materialfor the production of plastics, dyes, pharmaceuticals,syntans, and other products.
Phenol is one of the most versatile industrialorganic chemicals. It is the starting point formany diverse products used in the home andindustry. A partial list includes nylon, epoxyresins, surface active agents, synthetic detergents,plasticizers, antioxidants, lube oil additives,phenolic resins (with formaldehyde, furfural,and so on), cyclohexanol, adipic acid,polyurethanes, aspirin, dyes, wood preservatives,herbicides, drugs, fungicides, gasolineadditives, inhibitors, explosives, and pesticides.
Safety
Phenol is highly corrosive and toxic, the main effects being on the central nervous system. The lethal human oral dose is estimated to be 1 g for an adult.Phenol is absorbed from the gastrointestinal tract, skin, and mucous membranes, and is metabolized to phenylglucuronide and phenyl sulfate, which are excreted in the urine.
Although there are a number of reports describing the toxic effects of phenol, these largely concern instances of accidental poisoning or adverse reactions during its use as a therapeutic agent.Adverse reactions associated with phenol used as a preservative are less likely owing to the smaller quantities that are used; however, it has been suggested that the body burden of phenol should not exceed 50 mg in a 10-hour period.This amount could be exceeded following administration of large volumes of phenolpreserved medicines.
LD50 (mouse, IV): 0.11 g/kg
LD50 (mouse, oral): 0.3 g/kg
LD50 (rabbit, skin): 0.85 g/kg
LD50 (rat, skin): 0.67 g/kg
LD50 (rat, oral): 0.32 g/kg
LD50 (rat, SC): 0.46 g/kg
잠재적 노출
Phenol is used as a pharmaceutical, in the production of fertilizer; coke, illuminating gas; lampblack, paints, paint removers; rubber, asbestos goods; wood preservatives; synthetic resins; textiles, drugs, pharmaceutical preparations; perfumes, bakelite, and other plastics (phenolformaldehyde resins); polymer intermediates (caprolactam, bisphenol-A and adipic acid). Phenol also finds wide use as a disinfectant and veterinary drug.Carcinogenicity
Phenol had been investigated for carcinogenicity in animals by the oral and dermal routes. IARC and IRIS determined that animal human evidence for carcinogenicity was inadequate.저장
When exposed to air and light, phenol turns a red or brown color, the color being influenced by the presence of metallic impurities. Oxidizing agents also hasten the color change. Aqueous solutions of phenol are stable. Oily solutions for injection may be sterilized in hermetically sealed containers by dry heat. The bulk material should be stored in a well-closed, light-resistant container at a temperature not exceeding 15°C.운송 방법
UN1671 Phenol, solid, Hazard Class: 6.1; Labels: 6.1-Poisonous materials. UN2312 Molten phenol, Hazard Class: 6.1; Labels: 6.1-Poisonous materials. UN2821 Phenol solutions, Hazard Class: 6.1; Labels: 6.1-Poisonous materials.비 호환성
Phenol, available in solid or liquid form, is colorless to light pink and has a sweet aromatic odor. It is stable under normal conditions of storage and use. The liquid and vapor are combustible. Phenol is incompatible with strong oxidizing agents, calcium hypochlorite, halogens, halogenated compounds, aluminum chloride, and nitrobenzene. Hot phenol can attack aluminum, lead, magnesium and zinc. It can react exothermally with peroxymonosulfuric acid, sodium nitrate, 1,3-butadiene and boron trifluoride diethyl ether. When phenol is heated to decomposition (ca. 715 °C), decomposition products include carbon monoxide and carbon dioxide.https://www.cdc.gov/niosh/npg/npgd0493.html
http://www51.honeywell.com/sm/common/documents/Public_Risk_Summary_-_GPS0075_Phenol_Dec_2012.pdf
폐기물 처리
Consult with environmental regulatory agencies for guidance on acceptable disposal practices. Generators of waste containing this contaminant (≥100 kg/mo) must conform with EPA regulations governing storage, transportation, treatment, and waste disposal. Incineration.주의 사항
Acute poisoning of phenol by ingestion, inhalation or skin contact may lead to death. Phenol is readily absorbed through the skin. It is highly toxic by inhalation. It is corrosive and causes burns and severe irritation effects. During use and handling of phenol, occupational workers should be very careful. Workers should use protective clothing, rubber boots, and goggles to protect the eyes from vapors and spillage.Regulatory Status
Included in the FDA Inactive Ingredients Database (injections). Included in medicines licensed in the UK. Included in the Canadian List of Acceptable Non-medicinal Ingredients.페놀 준비 용품 및 원자재
원자재
이소프로필(4-)아닐린
액화석유가스
아이소프로투론
큐멘
Phenol crude
프로필렌
황산 나트륨
아황산나트륨
이산화 황
Benzenesulfonic acid sodium salt
삼산화황
콜타르 증류액
벤젠
큐멘하이드로퍼옥사이드
수산화나트륨
유리솜
석유 에테르
황산
준비 용품
4-클로로-o-톨옥시 아세트산
3-CHLORO-4-PHENOXYANILINE
SODIUM PHENOLATE TRIHYDRATE
Tetrabromophenol Blue
비사코딜
3-Phenoxypropionic acid
2,4,6-트리-터셔리-부틸페놀
Dispersing agent S
비스페놀 A 디메타크릴산염
4-NITROPHENYL PHENYL ETHER
2,4,6-트리클로로페놀
프탈산 디페닐
4-Phenoxybutanoic acid
4-Chromanone
2-페녹시프로피온산
인산트리스(이소프로필페닐)
스테아릴-β-(3,5-디-t-부틸-4-하이드록시페닐)프로피오네이트
2,4-디-t-부틸페놀
트리스(2,4-디-터셔리-부틸페닐)포스파이트
3-PHENOXYANILINE
4'-Phenoxyacetophenone
펜타클로로펜산 나트륨
High temperature levelling agent BOF
p-코우마릭 산
6-PHENOXYNICOTINONITRILE
Roxatidine acetate
Fenpropathrin
파라-3차-부틸페놀-포름알데히드 수지
Sulphur Indanthrene Blue RNX
3-AMINO-1-PHENOXY-2-PROPANOL HYDROCHLORIDE
Acid Red 111
4'-Hydroxyvalerophenone
Antioxidant T502
synthetic tanning agent No.28
Seratrodast
4-히드록시만데릭산 일수화물
Disperse purple H-FRL
크산텐
나이트로펜(나이트로펜)
Adhesive 706
페놀 공급 업체
글로벌( 780)공급 업체
| 공급자 | 전화 | 이메일 | 국가 | 제품 수 | 이점 |
|---|---|---|---|---|---|
| Shandong Quark Chemical Co., Ltd. | +8613325218432 |
sales@quarkchem.com | China | 57 | 58 |
| Shandong Yanshuo Chemical Co., Ltd. | +86-18678179670 +86-18615116763 |
sales@yanshuochem.com | China | 101 | 58 |
| PT CHEM GROUP LIMITED | +86-85511178 +86-85511178 |
peter68@ptchemgroup.com | China | 35453 | 58 |
| Hebei Jingbo New Material Technology Co., Ltd | +8619931165850 |
hbjbtech@163.com | China | 1000 | 58 |
| Yujiang Chemical (Shandong) Co.,Ltd. | +86-17736087130 +86-18633844644 |
catherine@yjchem.com.cn | China | 355 | 58 |
| ZHEJIANG JIUZHOU CHEM CO., LTD | +86-0576225566889 +86-13454675544 |
admin@jiuzhou-chem.com;jamie@jiuzhou-chem.com;alice@jiuzhou-chem.com | China | 19949 | 58 |
| Shanghai UCHEM Inc. | +862156762820 +86-13564624040 |
sales@myuchem.com | China | 6709 | 58 |
| Capot Chemical Co.,Ltd. | 571-85586718 +8613336195806 |
sales@capotchem.com | China | 29797 | 60 |
| Henan Tianfu Chemical Co.,Ltd. | +86-0371-55170693 +86-19937530512 |
info@tianfuchem.com | China | 21689 | 55 |
| ATK CHEMICAL COMPANY LIMITED | +undefined-21-51877795 |
ivan@atkchemical.com | China | 32686 | 60 |