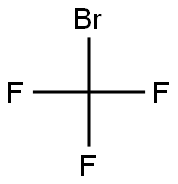
BROMOTRIFLUOROMETHANE
- CAS No.
- 75-63-8
- Chemical Name:
- BROMOTRIFLUOROMETHANE
- Synonyms
- CF3Br;halon-1301;CBrF3;H-1301;halon1301;Bromotrifluromethane;r13b1;f13b1;fe1301;fc13b1
- CBNumber:
- CB9430336
- Molecular Formula:
- CBrF3
Lewis structure

- Molecular Weight:
- 148.91
- MDL Number:
- MFCD00000370
- MOL File:
- 75-63-8.mol
| Melting point | -168°C |
|---|---|
| Boiling point | -58°C |
| Density | 1,58 g/cm3 |
| vapor pressure | >760 at 20 °C (NIOSH, 1997) |
| refractive index | 1.2380 |
| solubility | Soluble in chloroform (Weast, 1986) and many other solvents, particularly chlorinated hydrocarbons. |
| form | A gas |
| color | Colorless gas with an ether-like odor |
| Water Solubility | 0.03 wt % at 20 °C (NIOSH, 1997) |
| Henry's Law Constant | (atm?m3/mol): 0.500 at 25 °C (Hine and Mookerjee, 1975) |
| Exposure limits | NIOSH REL: TWA 1,000 ppm (6,100 mg/m3), IDLH 40,000 ppm; OSHA PEL: TWA 1,000 ppm. |
| CAS DataBase Reference | 75-63-8(CAS DataBase Reference) |
| FDA UNII | 52231LCA7R |
| EPA Substance Registry System | Halon 1301 (75-63-8) |
SAFETY
Risk and Safety Statements
| Symbol(GHS) |   GHS07,GHS04 |
|---|---|
| Signal word | Warning |
| Hazard statements | H280-H336-H420 |
| Precautionary statements | P410+P403-P502-P261-P271-P304+P340-P312-P403+P233-P405-P501 |
| Risk Statements | 20 |
| Safety Statements | 23-36/37/39-38 |
| RIDADR | 1009 |
| OEB | A |
| OEL | TWA: 1000 ppm (6100 mg/m3) |
| DOT Classification | 2.2 (Nonflammable gas) |
| HazardClass | 2.2 |
| Hazardous Substances Data | 75-63-8(Hazardous Substances Data) |
| Toxicity | LC50 (inhalation) for mice 381 gm/m3, rats 416 gm/m3 (quoted, RTECS, 1985). |
| IDLA | 40,000 ppm |
BROMOTRIFLUOROMETHANE Chemical Properties,Uses,Production
Chemical Properties
Trifluorobromomethane is a colorless gas with a slight ethereal odor. Shipped as a liquefied compressed gas.
Uses
Fire extinguishing agent; refrigerant.
Uses
Used as fire extinguishing agent for oil, electrical equipment, organic solvent, natural gas and a variety of organics, especially for important military and civilian sites.
Synthesis Reference(s)
Journal of the American Chemical Society, 68, p. 968, 1946 DOI: 10.1021/ja01210a017
General Description
A colorless, odorless gas at room conditions Shipped as a liquid confined under its own vapor pressure. Noncombustible. Nontoxic but can asphyxiate by the displacement of air. Contact with the unconfined liquid can cause frostbite by evaporative cooling. Exposure of the container to prolonged heat or fire can cause BROMOTRIFLUOROMETHANE to rupture violently and rocket.
Air & Water Reactions
Slightly soluble in water.
Reactivity Profile
BROMOTRIFLUOROMETHANE may react with aluminum to produce substantial heat. Other halogenated hydrocarbons, such as fluorotrichloromethane, dichlorodifluoromethane, chlorodifluoromethane, tetrafluoromethane produce sufficient heat in this way to melt aluminum pieces. The vigor of the reaction appears to depend on the degree of fluorination and the vapor pressure [Chem. Eng. News 39(27):44 1961].
Health Hazard
Vapors may cause dizziness or asphyxiation without warning. Vapors from liquefied gas are initially heavier than air and spread along ground. Contact with gas or liquefied gas may cause burns, severe injury and/or frostbite. Fire may produce irritating, corrosive and/or toxic gases.
Fire Hazard
Some may burn but none ignite readily. Containers may explode when heated. Ruptured cylinders may rocket.
Safety Profile
Wildly toxic by inhalation. Incompatible with aluminum. When heated to decomposition it emits toxic fumes of Fand Br-. See also BROMIDES and FLUORIDES.
Potential Exposure
This material is used as a fire extinguishing agent, a chemical intermediate, and as a refrigerant.
Shipping
UN1009 Bromotrifluoromethane or Refrigerant gas, R-13B, Hazard Class: 2.2; Labels: 2.2-Nonflammable gas. Cylinders must be transported in a secure upright position, in a well-ventilated truck. Protect cylinder and labels from physical damage. The owner of the compressed gas cylinder is the only entity allowed by federal law (49CFR) to transport and refill them. It is a violation of transportation regulations to refill compressed gas cylinders without the express written permission of the owner.
Purification Methods
Purify the gas by passing it through a tube containing P2O5 on glass wool into a vacuum system where it is frozen out in a quartz tube and degassed by a cycles of freezing, evacuating and thawing. [Beilstein 1 III 83, 1 IV 73.]
Incompatibilities
Keep away from chemically active metals, such as calcium, powdered aluminum; zinc, magnesium. Attacks some plastics, rubber, and coatings.
Waste Disposal
Return refillable compressed gas cylinders to supplier. Incineration, preferably after mixing with another combustible fuel. Care must be exercised to assure complete combustion to prevent the formation of phosgene. An acid scrubber is necessary to remove the halo acids produced.
BROMOTRIFLUOROMETHANE Preparation Products And Raw materials
Raw materials
Preparation Products
1of2