THIOMETON
- CAS No.
- 640-15-3
- Chemical Name:
- THIOMETON
- Synonyms
- m81;M 81;ebicid;ekatim;Medrin;san230;Veltin;EKATIN;Nimeton;SAN 230
- CBNumber:
- CB0731014
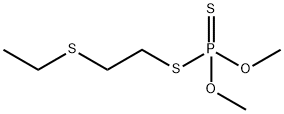
- Molecular Formula:
- C6H15O2PS3
- Molecular Weight:
- 246.35
- MOL File:
- 640-15-3.mol
- Modify Date:
- 2023/5/15 10:42:51
| Melting point | <25℃ |
|---|---|
| Boiling point | 110°C |
| Density | 1.209 g/cm3 (20℃) |
| vapor pressure | 3.99×10-2 Pa (20 °C) |
| refractive index | 1.568 (589.3 nm 20℃) |
| Flash point | 70 °C |
| storage temp. | 2-8°C |
| solubility | soluble in No data available |
| Water Solubility | 200 mg l-1(25 °C) |
| form | liquid |
| CAS DataBase Reference | 640-15-3 |
| EPA Substance Registry System | Thiometon (640-15-3) |
SAFETY
Risk and Safety Statements
| Symbol(GHS) |  GHS06 |
|---|---|
| Signal word | Danger |
| Hazard statements | H301-H312 |
| Precautionary statements | P264-P270-P301+P310-P321-P330-P405-P501-P280-P302+P352-P312-P322-P363-P501 |
| Hazard Codes | T,N |
| Risk Statements | 21-25-20/21-66-65-51/53-40 |
| Safety Statements | 36/37-45-62-61 |
| RIDADR | 3018 |
| HazardClass | 6.1(a) |
| PackingGroup | II |
THIOMETON Chemical Properties,Uses,Production
Description
Thiometon is the thion (P=S) analog of demeton-Smethyl. It is a colorless oil, bp 104 ?C/0.3 mm Hg, vp 39.9 mPa (20 ?C). The water solubility is 200 mg/L (27 ?C). It is highly soluble inmost organic solvents except alkanes. Log Kow = 3.15. It is hydrolyzed in alkaline and acidic media; DT50 (25 ?C) values at pH 3, 6, and 9 are 25, 27, and 17 d, respectively.
Uses
Thiometon is used to control sucking insects and mites on a wide range of crops.
Safety Profile
Poison by ingestion, skin contact, inhalation, and intravenous routes. Mutation data reported. A skin and severe eye irritant. A cholinesterase inhibitor. When heated to decomposition it emits very toxic fumes of POx and SOx. See also ESTERS and PARATHION
Metabolic pathway
Thiometon is the thion (P=S) analogue of demeton-S-methyl. Consequently, many of the biotransformations of hometon are similar to those of demeton-S-methyl to which it is oxidised in animals. It is also the dimethyl analogue of disulfoton. However, thiooxidation in soil and plants appears to be more facile than oxidative desulfuration to the oxon so that its metabolism is analogous to phorate and disulfoton in that the compound is first thiooxidised to thiometon sulfoxide and sulfone which are oxidatively desulfurated to oxydemeton-S-methyl and demeton-S-methylsulfon respectively.





