Tranexamic Acid
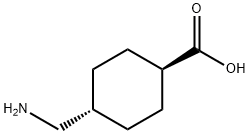
- CAS No.
- 1197-18-8
- Chemical Name:
- Tranexamic Acid
- Synonyms
- Tranexamic;(1r,4r)-4-(aminomethyl)cyclohexane-1-carboxylic acid;AMCA;cyklokapron;(1r,4r)-4-(aMinoMethyl)cyclohexanecarboxylic acid;TRANS-4-(AMINOMETHYL)CYCLOHEXANECARBOXYLIC ACID;amstat;transamin;Tranexamic Acid D2;TIMTEC-BB SBB006715
- CBNumber:
- CB1117189
- Molecular Formula:
- C8H15NO2
- Molecular Weight:
- 157.21
- MOL File:
- 1197-18-8.mol
- MSDS File:
- SDS
- Modify Date:
- 2024/7/14 20:44:35
| Melting point | >300 °C (lit.) |
|---|---|
| Boiling point | 281.88°C (rough estimate) |
| Density | 1.0806 (rough estimate) |
| vapor pressure | 1.72hPa at 25℃ |
| refractive index | 1.4186 (estimate) |
| storage temp. | 2-8°C |
| solubility | Freely soluble in water and in glacial acetic acid, practically insoluble in acetone and in ethanol (96 per cent). |
| pka | pKa 4.3 (Uncertain);10.6 (Uncertain) |
| form | Crystalline Powder |
| color | White |
| Water Solubility | 1g/6ml |
| Merck | 14,9569 |
| BRN | 2207452 |
| Stability | Hygroscopic |
| InChIKey | GYDJEQRTZSCIOI-LJGSYFOKSA-N |
| CAS DataBase Reference | 1197-18-8 |
SAFETY
Risk and Safety Statements
| Symbol(GHS) |  GHS07 |
|||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Signal word | Warning | |||||||||
| Hazard statements | H315-H319-H335 | |||||||||
| Precautionary statements | P261-P264-P271-P280-P302+P352-P305+P351+P338 | |||||||||
| Hazard Codes | Xi | |||||||||
| Risk Statements | 36/37/38 | |||||||||
| Safety Statements | 26-36-37/39 | |||||||||
| WGK Germany | 2 | |||||||||
| RTECS | GU8400000 | |||||||||
| HazardClass | IRRITANT | |||||||||
| HS Code | 29224999 | |||||||||
| Toxicity | LD50 in mice, rats (mg/kg): 1500, 1200 i.v. (Melander) | |||||||||
| NFPA 704 |
|
Tranexamic Acid price More Price(7)
| Manufacturer | Product number | Product description | CAS number | Packaging | Price | Updated | Buy |
|---|---|---|---|---|---|---|---|
| Sigma-Aldrich(India) | 857653 | trans-4-(Aminomethyl)cyclohexanecarboxylic acid 97% | 1197-18-8 | 10G | ₹4546.5 | 2022-06-14 | Buy |
| Sigma-Aldrich(India) | 857653 | trans-4-(Aminomethyl)cyclohexanecarboxylic acid 97% | 1197-18-8 | 50G | ₹14689.53 | 2022-06-14 | Buy |
| Sigma-Aldrich(India) | PHR1812 | Tranexamic acid Pharmaceutical Secondary Standard; Certified Reference Material | 1197-18-8 | 500MG | ₹16215.85 | 2022-06-14 | Buy |
| Sigma-Aldrich(India) | 857653 | trans-4-(Aminomethyl)cyclohexanecarboxylic acid 97% | 1197-18-8 | 250G | ₹49372.83 | 2022-06-14 | Buy |
| TCI Chemicals (India) | A0236 | trans-4-(Aminomethyl)cyclohexanecarboxylic Acid min. 98.0 % | 1197-18-8 | 5G | ₹2100 | 2022-05-26 | Buy |
Tranexamic Acid Chemical Properties,Uses,Production
Description
Tranexamic acid is a derivative of aminomethylbenzoic acid, and a kind of antifibrinolytic drugs to stop bleeding. The hemostasis mechanism of tranexamic acid is similar to aminocaproic acid and aminomethylbenzoic acid, but the effect is stronger. The strength is 7 to 10 times of aminocaproic acid, 2 times of aminomethylbenzoic acid, but toxicity is similar.
The chemical structure of tranexamic acid is similar to lysine, competitive inhibition of plasmin original in fibrin adsorption, to prevent their activation, protection fiber protein not to degrade by plasmin and dissolve, eventually achieve hemostasis. Applicable in the treatment of acute or chronic, localized or systemic primary fiber fibrinolytic hyperthyroidism caused by bleeding, such as obstetric hemorrhage, renal hemorrhage, hemorrhage of hypertrophy of the prostate, hemophilia, pulmonary tuberculosis hemoptysis, stomach bleeding, after operation of liver, lung, spleen and other viscera hemorrhage; also can be used in surgery when abnormal bleeding etc..
Clinical tranexamic acid has effect significantly to insect bites disease, dermatitis and eczema, simple purpura, chronic urticaria, artificial sex urticaria, toxic eruption and eruption. And also has a certain effect on erythroderma, scleroderma, systemic lupus erythematosus (SLE), Erythema multiforme, shingles and alopecia areata. Treatment of hereditary angioedema effect is also good. In the treatment of Chloasma, general medicine is effective about 3 weeks, markedly effective 5 weeks, a course of 60 days. Given orally in doses of 0.25 ~ 0.5 g, a day 3 ~ 4 times. A few patients can nausea, fatigue, pruritus, abdominal discomfort, and diarrhea side effects after withdrawal symptoms disappear.
Chemical Properties
Tranexamic acid is a White or almost white, crystalline powder. It is freely soluble in water and in glacial acetic acid and is very slightly soluble in ethanol and practically insoluble in ether.
Uses
Fibrinolysis, the cleavage of fibrin by plasmin, is a normal step in the dissolution of fibrin clots after wound repair. Tranexamic acid is an inhibitor of fibrinolysis that blocks the interaction of plasmin with fibrin (IC50 = 3.1 μM). It is a lysine mimetic that binds the lysine binding site in plasmin. Antifibrinolytic agents have value when fibrinolytic activity is abnormally high or when coagulation is impaired.
Definition
ChEBI: Tranexamic acid is a monocarboxylic acid. It has a role as an antifibrinolytic drug and a hematologic agent. It is functionally related to a cyclohexanecarboxylic acid.
General Description
Tranexamic acid is an antifibrinolytic agent and is commonly used for heavy menstrual bleeding.
Mechanism of action
Tranexamic acid is a synthetic lysine amino acid derivative, which diminishes the dissolution of hemostatic fibrin by plasmin. In the presence of tranexamic acid, the lysine receptor binding sites of plasmin for fibrin are occupied, preventing binding to fibrin monomers, thus preserving and stabilizing fibrin’s matrix structure. This agent has a longer half-life, is approximately ten times more potent, and is less toxic than aminocaproic acid, which possesses similar mechanisms of action.
Side effects
Tranexamic acid is mostly well-tolerated.
Common
nausea (upset stomach)
vomiting (throwing up)
diarrhea
headache
Occasional
dizziness
giddiness
vision changes
Rare
stroke
blood clots in undesired areas
deep vein thrombosis
Pharmacokinetics
Tranexamic acid 1 g was given intravenously to three healthy volunteers. Plasma concentrations decayed in three monoexponential phases. Most elimination occurred during the first eight hours, giving an apparent elimination half-life of approximately two hours. Plasma clearance ranged between 110-116 ml/min. The urinary recovery of tranexamic acid exceeded 95% of the dose. The oral bioavailability of tranexamic acid, calculated from 24-hour urinary excretion after oral and intravenous administration, was 34% of the dose[1].
References
Intravenous use of tranexamic acid reduces postoperative blood loss in total knee arthroplasty DOI:10.1007/s00402-014-2081-x
www.childrensmn.org
dermnetnz.org
Tranexamic acid for the prevention and treatment of postpartum haemorrhage. DOI:10.1097/01.aoa.0000552886.12061.3c
Sittig's Pharmaceutical Manufacturing Encyclopedia
The Renal Drug Handbook
Synthesis of Essential Drugs (2006, Elsevier) - libgen.lc
Spectrophotometric and spectrofluorimetric methods for the determination of tranexamic acid in pharmaceutical formulation. DOI:10.1248/CPB.55.364
Determination of plasma tranexamic acid using cation-exchange high-performance liquid chromatography with fluorescence detection. DOI:10.1016/S0378-4347(00)84573-9
[1] A Pilbrant, J Vessman, M Schannong. “Pharmacokinetics and bioavailability of tranexamic acid.” European Journal of Clinical Pharmacology 20 1 (1981): 65–72.
Tranexamic Acid Preparation Products And Raw materials
Raw materials
1of2
chevron_rightPreparation Products
| Supplier | Tel | Country | ProdList | Advantage | Inquiry |
|---|---|---|---|---|---|
| SIMSON PHARMA LIMITED | +91-8291074273 | Shanghai, India | 228 | 58 | Inquiry |
| CHEMXTREE STANDARDS | +919872732255 | Punjab, India | 96 | 58 | Inquiry |
| GGC Chemicals And Pharmaceuticals Pvt Ltd | +919824336772 | Gujarat, India | 14 | 58 | Inquiry |
| Medi Pharma Drug House | +919930911911 | Mumbai, India | 143 | 58 | Inquiry |
| CNS CHEMICALS | +91-9381957258 +91-9381957258 | Telangana, India | 354 | 58 | Inquiry |
| Kawman Pharma (A Division of K P Manish Global Ingredients Pvt Ltd) | +919043474090 | Tamil Nadu, India | 16 | 58 | Inquiry |
| Malladi Drugs AND Pharmaceuticals Limited | +91-9004653310 +91-9003056437 | Tamil Nadu, India | 69 | 58 | Inquiry |
| Venture Pharmaceuticals pvt ltd | +91-9173909075 +91-9173909075 | Gujarat, India | 45 | 58 | Inquiry |
| Shilpa Medicare Limited (SML) | +91-8532238704 +91-9320649838 | Karnataka, India | 61 | 58 | Inquiry |
| PROTECH TELELINKS | +91-8571891912 +91-9855060837 | Himachal Pradesh, India | 62 | 58 | Inquiry |
Related articles
- Tranexamic Acid: Pharmacological Effects and Adverse Effects
- Tranexamic acid inhibits plasmin formation, enhances hemostasis, and modulates melanocyte activity, with adverse effects inclu....
- May 30,2024
- Medical effects of tranexamic acid
- Tranexamic acid, also known as tranexamic acid, tranexamic acid, hemostatic acid, molecular formula is C8H15NO2
- Mar 30,2022
- Side effects of Tranexamic acid
- Tranexamic acid (TXA) is a medication used to treat or prevent excessive blood loss from major trauma, postpartum bleeding, su....
- Mar 8,2022
1197-18-8(Tranexamic Acid)Related Search:
1of4
chevron_right




