ChemicalBook > Product Catalog >Analytical Chemistry >Standard >Pharmaceutical Impurity Reference Standards >Cabozantinib Impurity EXA
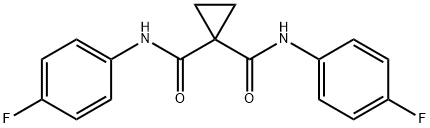
Cabozantinib Impurity EXA
- CAS No.
- 1431468-36-8
- Chemical Name:
- Cabozantinib Impurity EXA
- Synonyms
- Cabozantinib Impurity 07;Cabozantinib impurities8;Cabozantinib Impurity EXA;N,N'-bis(4-fluorophenyl)cyclopropane-1,1-dicarboxamide ;1,1-Cyclopropanedicarboxamide, N,N'-bis(4-fluorophenyl)-;N,N'-bis(4-fluorophenyl)cyclopropane-1,1-dicarboxamide (Cabozantinib Impurity);N,N'-bis(4-fluorophenyl)cyclopropane-1,1-dicarboxamide? (Cabozantinib Impurity)
- CBNumber:
- CB55855325
- Molecular Formula:
- C17H14F2N2O2
- Molecular Weight:
- 316.3
- MOL File:
- 1431468-36-8.mol
- Modify Date:
- 2022/12/21 16:56:50
Cabozantinib Impurity EXA Preparation Products And Raw materials
Raw materials
Preparation Products
Cabozantinib Impurity EXA Suppliers
Global( 34)Suppliers
| Supplier | Tel | Country | ProdList | Advantage | Inquiry |
|---|---|---|---|---|---|
| Pharmaffiliates Analytics and Synthetics P. Ltd | +91-172-5066494 | Haryana, India | 6773 | 58 | Inquiry |
| ATK CHEMICAL COMPANY LIMITED | +undefined-21-51877795 | China | 32760 | 60 | Inquiry |
| ANHUI WITOP BIOTECH CO., LTD | +8615255079626 | China | 23556 | 58 | Inquiry |
| Shenzhen Roark Pharma-Tec Co., Ltd. | 13128912434 | China | 2579 | 58 | Inquiry |
| LGC Science (Shanghai) Ltd. | 17717235263 | China | 11572 | 58 | Inquiry |
| Shanghai Zerui Biotechnology Co., Ltd. | 021-54580889 18321124657 | China | 10614 | 58 | Inquiry |
| Shanghai Send Pharmaceutical Technology Co., Ltd. | 021-58088081 Q1224289022 | China | 3031 | 55 | Inquiry |
| Hubei Ruinuo Pharmaceutical Technology Co., Ltd | 13165688766 | China | 5000 | 58 | Inquiry |
| Shenzhen Qingtian Biotechnology Co, Ltd. | 18823167005 18162969855 | China | 2628 | 58 | Inquiry |
| Beijing Comparison Pharmaceutical Technology Co., Ltd | 010-010-52878169 15801002657 | China | 4783 | 58 | Inquiry |
1431468-36-8(Cabozantinib Impurity EXA)Related Search:
4-Aminoveratrole
Cabozantinib Impurity 5
N-(4-fluorophenyl)-N-(4-hydroxyphenyl)cyclopropane-1,1-dicarboxamide
4-chloro-6,7-dimethoxyquinoline
Cabozantinib impurity DX2
1-(4-Fluorophenylcarbamoyl)cyclopropanecarboxylic acid
Cyclopropanecarboxylic acid, 1-[[[4-[[7-methoxy-6-(sulfooxy)-4-quinolinyl]oxy]phenyl]amino]carbonyl]-
1,1-Cyclopropanedicarboxamide,N-[4-[(6,7-dimethoxy-4-quinolinyl)oxy]phenyl]-N'-phenyl-
Quinoline, 6,7-dimethoxy-4-(4-nitrophenoxy)-
Cyclopropanecarboxylic acid, 1-[(phenylaMino)carbonyl]-
chevron_left
1of3
chevron_right
Cabozantinib Impurity EXA
1,1-Cyclopropanedicarboxamide, N,N'-bis(4-fluorophenyl)-
N,N'-bis(4-fluorophenyl)cyclopropane-1,1-dicarboxamide
N,N'-bis(4-fluorophenyl)cyclopropane-1,1-dicarboxamide? (Cabozantinib Impurity)
Cabozantinib impurities8
Cabozantinib Impurity 07
N,N'-bis(4-fluorophenyl)cyclopropane-1,1-dicarboxamide (Cabozantinib Impurity)
1431468-36-8





