Cadmium sulfide
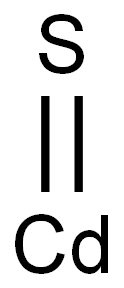
- CAS No.
- 1306-23-6
- Chemical Name:
- Cadmium sulfide
- Synonyms
- gsk;99%;pc108;capsebon;Yellow 37;C.I.77199;nci-c02711;nci-co2711;ferroyellow;greenockite
- CBNumber:
- CB6853020
- Molecular Formula:
- CdS
- Molecular Weight:
- 144.48
- MOL File:
- 1306-23-6.mol
- MSDS File:
- SDS
- Modify Date:
- 2024/5/20 14:48:35
| Melting point | 980°C (subl.) |
|---|---|
| Boiling point | 980°C (estimate) |
| Density | 4.82 g/mL at 25 °C(lit.) |
| Flash point | 4℃ |
| storage temp. | 2-8°C |
| solubility | Soluble in acid, very slightly soluble in ammonium hydroxide. |
| form | powder |
| color | Yellow to orange |
| Specific Gravity | 4.82 |
| Water Solubility | Insoluble |
| Crystal Structure | Cubic, Sphalerite Structure - Space Group F(-4)3m |
| Merck | 14,1628 |
| Solubility Product Constant (Ksp) | pKsp: 26.1 |
| Exposure limits |
ACGIH: TWA 0.01 mg/m3; TWA 0.002 mg/m3 NIOSH: IDLH 9 mg/m3 |
| CAS DataBase Reference | 1306-23-6(CAS DataBase Reference) |
| EPA Substance Registry System | Cadmium sulfide (1306-23-6) |
| Hardness, Mohs | 3.3 |
|---|---|
| Knoop Microhardness | 1250, N/mm2 |
SAFETY
Risk and Safety Statements
| Symbol(GHS) |    GHS07,GHS08,GHS09 |
|||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Signal word | Danger | |||||||||
| Hazard statements | H302-H341-H350-H361fd-H372-H410 | |||||||||
| Precautionary statements | P201-P202-P260-P273-P301+P312-P308+P313 | |||||||||
| Hazard Codes | T,N,F | |||||||||
| Risk Statements | 45-22-48/23/25-53-62-63-68-50/53-67-65-48/20/22-38-11 | |||||||||
| Safety Statements | 53-45-61-60-62-36/37-16 | |||||||||
| RIDADR | UN 3077 9/PG 3 | |||||||||
| WGK Germany | 3 | |||||||||
| RTECS | EV3150000 | |||||||||
| TSCA | Yes | |||||||||
| HazardClass | 6.1 | |||||||||
| PackingGroup | III | |||||||||
| NFPA 704 |
|
Cadmium sulfide price More Price(18)
| Manufacturer | Product number | Product description | CAS number | Packaging | Price | Updated | Buy |
|---|---|---|---|---|---|---|---|
| Sigma-Aldrich(India) | 217921 | Cadmium sulfide powder, 99.995% trace metals basis | 1306-23-6 | 20G | ₹15923.58 | 2022-06-14 | Buy |
| Sigma-Aldrich(India) | 208183 | Cadmium sulfide powder | 1306-23-6 | 100G | ₹3171.73 | 2022-06-14 | Buy |
| ALFA India | ALF-A14544-36 | Cadmium sulfide, 98% | 1306-23-6 | 500g | ₹15871 | 2022-05-26 | Buy |
| ALFA India | ALF-A14544-22 | Cadmium sulfide, 98% | 1306-23-6 | 100g | ₹3509 | 2022-05-26 | Buy |
| ALFA India | ALF-A14544-0E | Cadmium sulfide, 98% | 1306-23-6 | 2500g | ₹63594 | 2022-05-26 | Buy |
Cadmium sulfide Chemical Properties,Uses,Production
Chemical Properties
Cadmium sulfide is an odorless, crystalline, lemon yellow to orange solid.
Physical properties
Yellow to orange crystal; occurs as two polymorphs, hexagonal alpha form and cubic beta form; exhibits stable wurtzite structure at lower temperature, and zinc blende type structure at higher temperatures; the beta form converts to alpha form when heated at 750°C in sulfur atmosphere; sublimes at 980°C; practically insoluble in water (1.3 mg/L at 20°C); Ksp 3.6x10-29; dissolves in dilute mineral acids on heating or concentrated acids at ordinary temperatures (decomposes with liberation of H2S).
Occurrence
Cadmium sulfide occurs in nature as the mineral greenoktite. The compound is widely used in pigments for paints, baking enamels, ceramics and plastics. It imparts bright yellow to maroon, with strong retention of color and resistance to alkalis. It also is used in inks, phosphors, and fluorescent screens. Other applications of this compound are in photovoltaic and solar cells (for converting solar energy to electrical energy), photoconductors (in xerography), thin film transistors and diodes, rectifiers, scintillation counters, pyrotechnics, and smoke detectors.
Uses
cadmium sulfide is used as a colorant for paints and rubber; cadmium acetate is used in the production of craftware.
Production Methods
Cadmium sulfide may be prepared by the reaction between hydrogen sulfide and cadmiumvapor at 800 Cor by heating a mixture of cadmium or cadmium oxide with sulfur.
Definition
A native cadmium sulfide containing 77.7% cadmium. Ore of cadmium
Preparation
Cadmium sulfide may be prepared by precipitation from an aqueous solution of its soluble salts such as cadmium chloride or cadmium nitrate by passing hydrogen sulfide. The reactions may be carried out in acidic, neutral or alkaline solutions using various cadmium salts to obtain different crystal modifications as shown in the table below.
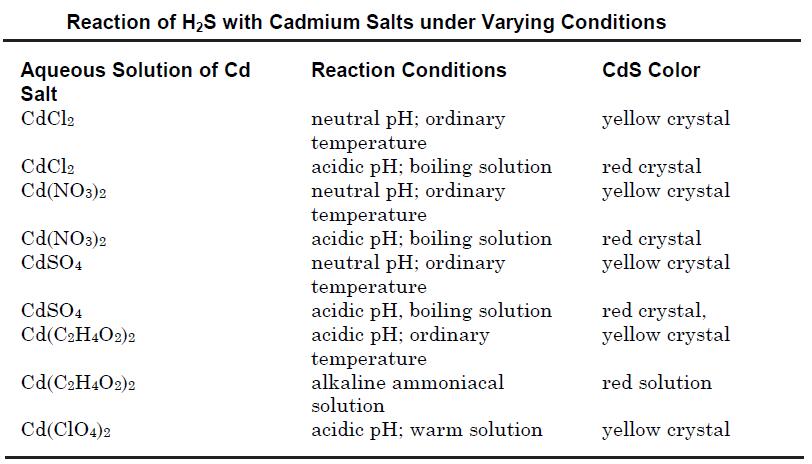
Cadmium sulfide also may be obtained by treatment of sodium or other alkali metal sulfide solution with that of a soluble cadmium salt. The compound also may be prepared by heating a mixture of cadmium or its oxide with sulfur at 800°C; or by the reaction of H2S with cadmium vapor at 800°C.
General Description
Natural occurrence: hawleyite (structural type of sphalerite) and greenockite (structural type of wurtzite)
Hazard
A confirmed carcinogen, highly toxic. See cadmium.
Safety Profile
Confirmed human carcinogen with experimental carcinogenic and tumorigenic data. Moderately toxic by ingestion and inhalation. Human mutation data reported. When heated to decomposition it emits very toxic fumes of Cd and SOx. See also CADMIUM COMPOUNDS and SULFIDES
Potential Exposure
Used in pigments; as an active ingredient in dandruff shampoos; making photoconductors, solar cells, and other electronic components.
Solubility in water
Cadmium sulfide is soluble in water, with a solubility of 13×10-5 g/100 ml H2O (18℃). CdS is soluble in acid.
Shipping
UN2570 Cadmium compounds, Hazard Class: 6.1; Labels: 6.1-Poisonous materials, Technical Name Required.
Structure and conformation
Cadmium sulfide takes two types of structure, zinc blend and wurtzite structures.
Cubic system that has a zinc-blend structure, with a lattice constant of a=0.582 nm and Cd– S=0.252 nm.
Hexagonal system that has a wurtzite structure with lattice constants of a=0.4136 nm, c=0.6713 nm and c/a=1.624, Cd–S=0.252 nm.
Incompatibilities
Contact with water or moisture releases poisonous hydrogen sulfide gas. Incompatible with oxidizers (chlorates, nitrates, peroxides, permanganates, perchlorates, chlorine, bromine, fluorine, etc.); contact may cause
Waste Disposal
Use a licensed professional waste disposal service to dispose of this material. All federal, state, and local environmental regulations must be observed. Consult with environmental regulatory agencies for guidance on acceptable disposal practices. Generators of waste containing this contaminant (≥100 kg/mo) must conform to EPA regulations governing storage, transportation, treatment, and waste disposa
Cadmium sulfide Preparation Products And Raw materials
| Supplier | Tel | Country | ProdList | Advantage | Inquiry |
|---|---|---|---|---|---|
| Scientific OEM | +91-22- 2343 7546 / 2341 3094 | New Delhi, India | 1996 | 38 | Inquiry |
| ALPHA CHEMIKA | +91-22-22061123 +91-22-66382501 | Mumbai, India | 1681 | 43 | Inquiry |
| S.K. CHEMICAL INDUSTRIES | +91-22-66370299 Ext 21 / 91-22-23437380 Ext 25 | New Delhi, India | 262 | 30 | Inquiry |
| Otto Chemie Pvt. Ltd. | +91 9820041841 | Mumbai, India | 5873 | 58 | Inquiry |
| Sisco Research Laboratories Pvt. Ltd. | +91-22-4268 5800 | Mumbai, India | 4317 | 58 | Inquiry |
| Alfa Aesar | 1 800 209 7001 | Maharashtra, India | 6913 | 58 | Inquiry |
| Triveni chemicals | 08048762458 | New Delhi, India | 6093 | 58 | Inquiry |
| LOBA CHEMIE PVT.LTD. | 91-22-6663 6699 | Mumbai, India | 3077 | 58 | Inquiry |
| Central Drug House(P) Ltd. | 91-11-49404040 | New Delhi, India | 6160 | 58 | Inquiry |
| Zama Chemical | 08048983954 | Mumbai, India | 217 | 58 | Inquiry |
| Supplier | Advantage |
|---|---|
| Scientific OEM | 38 |
| ALPHA CHEMIKA | 43 |
| S.K. CHEMICAL INDUSTRIES | 30 |
| Otto Chemie Pvt. Ltd. | 58 |
| Sisco Research Laboratories Pvt. Ltd. | 58 |
| Alfa Aesar | 58 |
| Triveni chemicals | 58 |
| LOBA CHEMIE PVT.LTD. | 58 |
| Central Drug House(P) Ltd. | 58 |
| Zama Chemical | 58 |
Related articles
- What is the crystal structure of Cadmium sulfide?
- CdS has two types of crystal structures, as shown above: (a) cubic zinc blende and (b) hexagonal wurtzite structures.
- May 20,2024
1306-23-6(Cadmium sulfide)Related Search:
1of4
chevron_right




