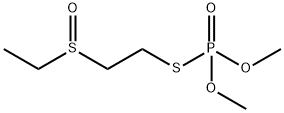
Oxydemeton-methyl
- CAS No.
- 301-12-2
- Chemical Name:
- Oxydemeton-methyl
- Synonyms
- r2170;B-21097;bay21097;ent24,964;bayer21097;NSC 370785;aimcosystox;metasystoxr;OxydeMeton M;metasystemox
- CBNumber:
- CB7115945
- Molecular Formula:
- C6H15O4PS2
- Molecular Weight:
- 246.28
- MOL File:
- 301-12-2.mol
- Modify Date:
- 2023/5/15 10:43:59
| Melting point | <-20℃ |
|---|---|
| Boiling point | 106 °C(Press: 0.01 Torr) |
| Density | 1.289 g/cm3 (20 ºC) |
| vapor pressure | 3.8×10-3 Pa (20°C) |
| refractive index | 1.5216 (589.3 nm 20℃) |
| storage temp. | 0-6°C |
| solubility | Chloroform (Sparingly), Ethyl Acetate (Slightly), Methanol (Slightly) |
| form | Oil |
| Water Solubility | Miscible |
| color | Colourless to Light Yellow |
| EPA Substance Registry System | Oxydemeton-methyl (301-12-2) |
SAFETY
Risk and Safety Statements
| Symbol(GHS) |   GHS06,GHS09 |
|---|---|
| Signal word | Danger |
| Hazard statements | H301+H311-H400 |
| Precautionary statements | P273-P280-P301+P310-P312 |
| Hazard Codes | T,N |
| Risk Statements | 24/25-50 |
| Safety Statements | 23-36/37-45-61 |
| RIDADR | 3018 |
| RTECS | TG1420000 |
| HazardClass | 6.1(a) |
| PackingGroup | II |
| Toxicity | LC50 (96-hour) for rainbow trout 4.0 mg/L (Walker, 1964); LC50 (24-hour) for rainbow trout and bluegill sunfish 10 mg/L (Hartley and Kidd, 1987); acute oral LD50 for rats 65–75 mg/kg (Hartley and Kidd, 1987), 30 mg/kg (RTECS, 1985). |
Oxydemeton-methyl Chemical Properties,Uses,Production
Description
Oxydemeton-methyl, i.e., demeton-S-methyl sulfoxide, is a colorless oil, bp 106 ?C/0.01 mm Hg, vp 3.8 mPa (20 ?C). It is miscible with water and soluble in most organic solvents, except petroleum ether. Log Kow = ?0.74(21 ?C). Oxydemeton-methyl is relatively stable in acidic media but hydrolyzed in alkalinemedia;DT50 values (22 ?C) at pH 4, 7, and 9 are 107, 46, and 2 d, respectively.
Chemical Properties
Oxydemeton methyl is a colorless to amber-colored liquid.
Oxydemeton methyl is relatively slowly hydrolyzed in acidic
media, but rapidly hydrolyzed in alkaline media.
Oxydemeton methyl is miscible with
water; readily soluble (10–100 g/
100 mL) in dichloromethane,
2-propanol, and toluene; and practically
insoluble (<1 g/100 mL) in n-hexane.
Uses
Oxydemeton-methyl is used to control sucking insects on fruit, vines, vegetables, cereals and ornamentals.
General Description
Clear amber liquid.
Air & Water Reactions
Water soluble. DEMETON-S-METHYL SULFOXIDE may be rapidly hydrolyzed by alkali .
Reactivity Profile
Organophosphates such as DEMETON-S-METHYL SULFOXIDE are susceptible to formation of highly toxic and flammable phosphine gas in the presence of strong reducing agents such as hydrides. Partial oxidation by oxidizing agents may result in the release of toxic phosphorus oxides.
Fire Hazard
DEMETON-S-METHYL SULFOXIDE is flammable.
Safety Profile
Poison by ingestion, skin contact, intravenous, and intraperitoneal routes. Human mutation data reported. When heated to decomposition it emits very toxic fumes of POx and SOx. See also other demeton entries.
Environmental Fate
Soil. The sulfoxide group is oxidized to the sulfone and oxidative and hydrolytic
cleavage of the side chain gives dimethylphosphoric and phosphoric acids (Hartley and
Kidd, 1987). Oxamyl was degraded by the microorganism Pseudomonas putida in a
laboratory study using cultured bacteria (Zeigler, 1980).
Plant. In asparagus, oxydemeton-methyl was converted to the corresponding sulfone
(Szeto and Brown, 1982).
Chemical/Physical. Oxydemeton-methyl can be converted to the corresponding sulfone
by hydrogen peroxide (Cremlyn, 1991). Emits toxic fumes of phosphorus and sulfur oxides
when heated to decomposition (Sax and Lewis, 1987).
Metabolic pathway
Oxydemeton-methyl is the primary thiooxidation metabolite of demeton- S-methyl. It is further oxidised rapidly to demeton-Smethylsulfon in all media. The major route of further metabolism of both sulfoxide and sulfone is via hydrolysis to the sulfoxide and sulfone thiols. Stage I1 metabolism of these thiol hydrolysis products proceeds via oxidation and conjugation and is different in soil, plants and animals. In plants the thiols are conjugated, whereas in animals they are mainly methylated and oxidised to sulfoxides, sulfones and sulfonic acids. Demethylation is an important route in mammals; however, the compound is sufficiently polar for much to be excreted in the urine unchanged. Demethylation has also been reported to occur in plants.
Metabolism
Almost 99% of orally administered oxydemeton-methyl to animals is excreted within 48 h in the urine. It is oxidized to the sulfone, followed by hydrolytic cleavage of the P?S bond. The thiol metabolites are conjugated or methylated. O-Demethylation is also an important degradation route both inmammals and plants. Oxydemeton-methyl is rapidly degraded in soils.
Oxydemeton-methyl Preparation Products And Raw materials
Related articles
- Oxydemeton-methyl Pesticides
- Oxydemeton-methyl is a metabolite of a previously marketed organophosphate pesticide, demeton-S-methyl.
- Oct 29,2021
301-12-2(Oxydemeton-methyl)Related Search:
1of4
chevron_right




