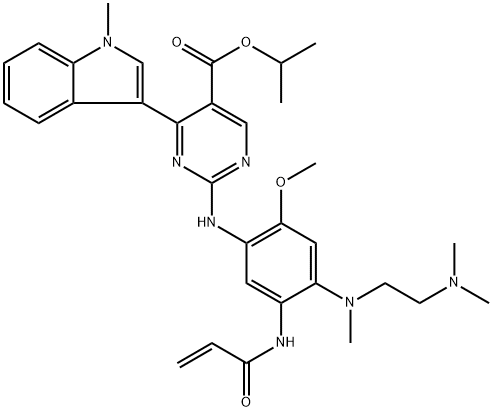
Mobocertinib
- CAS No.
- 1847461-43-1
- Chemical Name:
- Mobocertinib
- Synonyms
- Mobocertinib;Isopropyl 2-((5-acrylamido-4-((2-(dimethylamino)ethyl)(methyl)amino)-2-methoxyphenyl)amino)-4-(1-methyl-1H-indol-3-yl)pyrimidine-5-carboxylate;Mobocertinib (TAK788);5-Pyrimidinecarboxylic acid, 2-[[4-[[2-(dimethylamino)ethyl]methylamino]-2-methoxy-5-[(1-oxo-2-propen-1-yl)amino]phenyl]amino]-4-(1-methyl-1H-indol-3-yl)-, 1-methylethyl ester;TAK-788;TAK788 TAK-788;ak-788 Mobotinib;2H6]-Mobocertinib;AP32788, MOBOCERTINIB;TAK-788 (MOBOCERTINIB)
- CBNumber:
- CB74676602
- Molecular Formula:
- C32H39N7O4
- Molecular Weight:
- 585.71
- MOL File:
- 1847461-43-1.mol
- Modify Date:
- 2024/7/2 8:55:16
Mobocertinib Chemical Properties,Uses,Production
Description
Mobocertinib is derived from osimertinib, a second-generation TKI which exhibits only limited activity against several resistant EGFRex20ins mutants. Both inhibitors are structurally identical except that the pyrimidine ring of moboceritinib incorporates a snugly fitting isopropyl ester group that targets a previously unoccupied pocket, resulting in expanded coverage of EGFRex20ins mutations as well as improved selectivity over wildtype EGFR vs. osimertinib.
Uses
Mobocertinib (TAK-788) is an orally active and irreversible EGFR/HER2 inhibitor. Mobocertinib effectively inhibits oncogenic variants containing mutations that activate the EGFRex20ins, with selectivity superior to that of wild-type EGFR. Mobocertinib is FDA-approved for the treatment of patients with non-small cell lung cancer (NSCLC) who have a HER2 mutation or an EGFR mutation, including an exon 20 insertion mutation. Mobocertinib is approved by the FDA for the treatment of patients with non-small cell lung cancer (NSCLC) who have HER2 mutations or EGFR mutations, including exon 20 insertion mutations.
brand name
ExkivityTM
General Description
Class: receptor tyrosine kinase
Treatment: NSCLC with EGFR alterations
Oral bioavailability = 37%
Elimination half-life = 18 h
Biological Activity
In NSCLC cell lines, mobocertinib was highly active against common EGFR-activating mutations (exon 19 deletions and L858R) (IC50 = 1.3–4.0 nM) or with a gatekeeper T790M mutant (IC50 = 9.8 nM), as well as selective for wt EGFR (IC50 = 35 nM). In Ba/F3 cells, mobocertinib displayed activity against 14 EGFR mutations with IC50 values ranging from 2.7 to 22.5 nM, vs. 34.5 nM for wt EGFR. More specifically, mobocertinib inhibited all five variants of EGFRex20ins mutations with IC50 ranging from 4.3 nM to 22.5 nM.
target
EGFR, HER2-4
Metabolism
Moboceritinib undergoes CYP450-mediated metabolism to give two primary N-demethylated metabolites, AP32914 and AP32960, whose IC50 values are within 2-fold of mobocertinib for both wt and mutant EGFR. It is likely that these metabolites also contribute to the pharmacologic activity of mobocertinib. Interestingly, the typically labile isopropyl ester appears resistant to esterase-induced hydrolysis.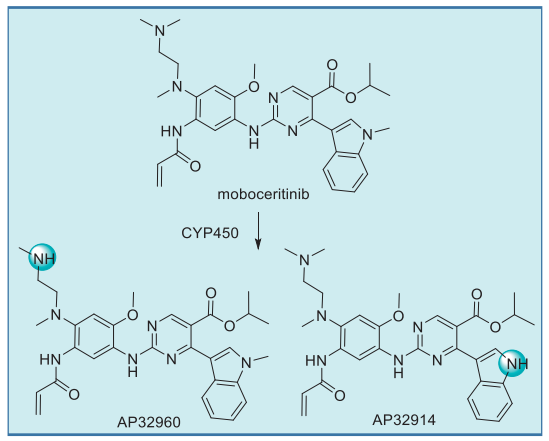
Mobocertinib Preparation Products And Raw materials
Raw materials
Preparation Products
Mobocertinib Suppliers
| Supplier | Tel | Country | ProdList | Advantage | Inquiry |
|---|---|---|---|---|---|
| Apicore Pharmaceuticals Pvt Ltd | +91-2662267177 +91-2662267166 | Gujarat, India | 181 | 58 | Inquiry |
| Aspen Biopharma Labs Pvt Ltd | +91-9248058660 +91-9248058662 | Telangana, India | 234 | 58 | Inquiry |
| CLEARSYNTH LABS LTD. | +91-22-45045900 | Hyderabad, India | 6351 | 58 | Inquiry |
| Zibo Hangyu Biotechnology Development Co., Ltd | +86-0533-2185556 +8617865335152 | China | 10978 | 58 | Inquiry |
| ATK CHEMICAL COMPANY LIMITED | +undefined-21-51877795 | China | 32760 | 60 | Inquiry |
| JIAXING REGEN CHEMICAL CO.,LTD | +8613385739570 | China | 226 | 58 | Inquiry |
| Career Henan Chemica Co | +86-0371-86658258 +8613203830695 | China | 30253 | 58 | Inquiry |
| Shanghai UCHEM Inc. | +862156762820 +86-13564624040 | China | 7034 | 58 | Inquiry |
| ANHUI WITOP BIOTECH CO., LTD | +8615255079626 | China | 23556 | 58 | Inquiry |
| Zhejiang J&C Biological Technology Co.,Limited | +1-2135480471 +1-2135480471 | China | 10522 | 58 | Inquiry |
Related articles
- Mobocertinib: Synthesis and Introduction
- Mobocertinib was approved as the first treatment for NSCLC with epidermal growth factor receptor (EGFR) exon 20 insertion muta....
- Jan 25,2024





