2-フェニルプロペン酸 化学特性,用途語,生産方法
外観
白色~ほとんど白色粉末~結晶
解説
2-フェニルプロペン酸融点106~107 ℃,沸点202~204 ℃(10 kPa).Ka 1.43×10-4.冷水に難溶,エタノールに可溶.融解を続けると二量化してtrans-1,2,3,4-テトラヒドロ-1-フェニル-1,4-ナフタレンジカルボン酸C18H16O4を与える.有機合成の原料となる.蒸気は刺激性があり,吸うと咳が出る.
森北出版「化学辞典(第2版)
製造
2-フェニルプロペン酸,トロパ酸を塩酸あるいは水酸化バリウムと加熱して脱水すると得られる.
化学的特性
White to Off-White Solid.
使用
Atropic acid is an Impurity of Ipratropium bromide and Atropine Sulfate. Atropic acid and β,β-dimethylatropic acid are plant growth regulators.
製造方法
The synthesis of atropic acid involves several steps. Firstly, tropic acid is oxidized with hot KMnO4 to form benzoic acid. Secondly, tropic acid reacted with HBr to produce C9H9O2Br. Thirdly, C9H9O2Br is reacted with alcoholic KOH to yield atropic acid, C9H8O2. Finally, atropic acid is catalytically hydrogenated to form hydratropic acid, C9H10O2.
反応性
Atropic acid is formed by the dehydration of tropic acid. Hence addition of water to atropic acid gives tropic acid.
Atropic acid, on oxidation yields benzoic acid. The formation of benzoic acid reveals that atropic acid and tropic acid contain atleast one benzene nucleus with a side chain containing carboxylic acid in their structure.
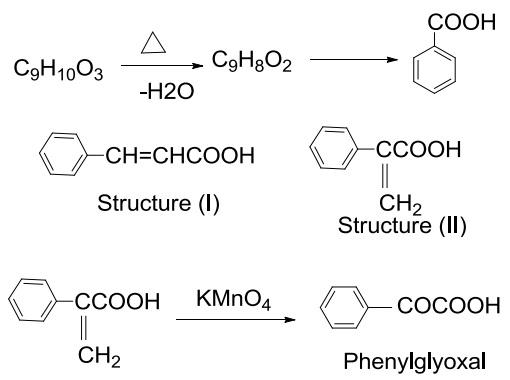
As atropic acid is an unsaturated acid it mean atropic acid may be either structure (I) or (II) .
Hence, the structure (II) is atropic acid which is confirmed by oxidation with KMnO4 to form phenylglyoxal.
2-フェニルプロペン酸 上流と下流の製品情報
原材料
準備製品