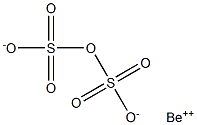
Beryllium Pyrosulfate
|
|
Beryllium Pyrosulfate 속성
안전
Beryllium Pyrosulfate C화학적 특성, 용도, 생산
개요
Beryllium pyrosulfate is not stable in aqueous solution and so must be prepared via a solid-state reaction. It would have the molecular formula of BeS2O7 and the molecular weight of 185.1401 g/mol. It could be prepared by the reaction involving “fuming” sulfuric acid (oleum) with beryllium sulfate:[Be(OH)]2SO4+(H2SO4)n ? 2BeS2O7+(n+2) H2O
The [Be(OH)]2SO4 salt is added to the oleum and allowed to dissolve in an excess of acid. The acid is heated to 150–175°C to drive off the water. This is an equivalent of using H2S2O7. The solution is allowed to equilibrate and crystals of the anhydrous salt appear. These are separated and allowed to dry. Beryllium disulfate is stable and may be heated to a red heat before it begins to decompose to the sulfate and SO3.
Beryllium Pyrosulfate 준비 용품 및 원자재
원자재
준비 용품
Beryllium Pyrosulfate 공급 업체
글로벌( 0)공급 업체
| 공급자 | 전화 | 이메일 | 국가 | 제품 수 | 이점 |
|---|