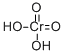
삼산화크롬무수물 C화학적 특성, 용도, 생산
개요
Chromic acid, CrO3, is composed of dark, purplish-red, odorless crystals that are soluble in water. The specific gravity is 2.7, which is heavier than water. It is a powerful oxidizing agent and may explode on contact with organic materials. Chromic acid is a poison, corrosive to the skin, and has a TLV of 0.05 mg/m3 of air. Chromic acid is a known human carcinogen. The four-digit UN identification number is 1463. The NFPA 704 designation is health 3, flammability 0, and reactivity 1. The white section at the bottom of the 704 diamond has an “oxy” prefix, indicating that it is an oxidizer.
화학적 성질
Chromic acid is a dark purplish-red odorless flakes or crystalline powder
용도
Chemicals (chromates, oxidizing agents, catalysts), chromium-plating intermediate, medicine
(caustic), process engraving, anodizing, ceramic
glazes, colored glass, metal cleaning, inks, tanning,
paints, textile mordant, etchant for plastics.
정의
The name is in common use, although the true
chromic acid, H2CrO4, exists only in solution.
일반 설명
Chromic acid is a dark purplish red solid, exists only in solution. The hydrate of chromiumoxide, it is used in electroplating baths. Chromic acid is soluble in water with the release of heat. The material itself is noncombustible but Chromic acid will accelerate the burning of combustible materials. Its solution is corrosive to metals and tissue.
공기와 물의 반응
Water soluble.
반응 프로필
A very powerful oxidizing agent, confirmed human carcinogen. Upon contact with reducing reagents Chromic acid can cause a violent explosion, in contact with organic matter Chromic acid may cause a violent oxidation leading to ignition. Dangerously reactive with acetone, alcohols, alkali metals (sodium, potassium), ammonia, arsenic, dimethylformamide, hydrogen sulfide, phosphorus, peroxyformic acid, pyridine, selenium, sulfur, and many other chemicals [Sax, 9th ed., 1996, p. 852]. When mixed with sulfuric acid for glass cleaning operations, used solution in closed bottle may explode due to internal pressure of carbon dioxide arising from contamination by carbon compounds [Bryson, W. R., Chem. Brit., 1975, 11, p. 377].
위험도
A human carcinogen. A poison. Corrosive
to skin. Powerful oxidizing agent, may explode on
contact with reducing agents, may ignite on contact with organic materials. Upper respiratory tract
irritant.
건강위험
Very irritating to eyes and respiratory tract. Ingestion causes severe gastrointestinal symptoms. Contact with eyes or skin causes burns; prolonged contact produces dermatitis (``chrome sores'').
화재위험
Behavior in Fire: Containers may explode
Safety Profile
Confirmed human
carcinogen. Poison by subcutaneous route.
Mutation data reported. A powerful
oxidzer. A powerful irritant of skin, eyes,
and mucous membranes. Can cause a
dermatitis, bronchoasthma, “chrome holes,”
damage to the eyes. Dangerously reactive.
Incompatible with acetic acid, acetic
anhydride, tetrahydronaphthalene, acetone,
alcohols, alkali metals, ammonia, arsenic,
bromine penta fluoride, butyric acid, n,ndimethylformamide, hydrogen sulfide,
peroxyformic acid, phosphorus, potassium
hexacyanoferrate, pyridme, selenium
잠재적 노출
n chromium plating; medicine, ceramic glazers, and paints.
운송 방법
UN1463 Chromium trioxide, anhydrous, Hazard Class: 5.1; Labels: 5.1-Oxidizer, 6.1-Poisonous materials, 8-Corrosive material. UN1755 (solution) Chromic acid, solid, Hazard class: 8; Labels: 8-Corrosive material.
비 호환성
A strong oxidizer. Aqueous solution is strongly acidic. Reacts with acetic acid, acetic anhydride, acetone, anthracene, chromous sulfide; diethyl ether; dimethyl formamide; ethanol, hydrogen sulfide; methanol, naphthalene, camphor, glycerol, potassium ferricyanide, pyridine, turpentine, combustibles; organics, and other easily oxidized materials (such as paper, wood, sulfur, aluminum, and plastics). Attacks metals in presence of moisture
폐기물 처리
Chemical reduction to chromium(III) can be followed by land fill disposal of the sludge.
삼산화크롬무수물 준비 용품 및 원자재
원자재
준비 용품