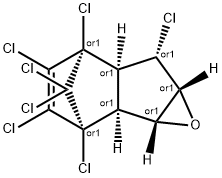
헵타클로르 에폭시드
|
|
헵타클로르 에폭시드 속성
- 녹는점
- 160-161.5℃
- 끓는 점
- 503.92°C (rough estimate)
- 밀도
- 1.7335 (rough estimate)
- 증기압
- 2.6(x 10-6 mmHg) at 20 °C (IARC, 1974)300(x 10-6 mmHg) at 30 °C (Nash, 1983)
- 굴절률
- 1.5000 (estimate)
- 인화점
- 11 °C
- 저장 조건
- APPROX 4°C
- 용해도
- 클로로포름(약간 용해됨), 에틸아세테이트(약간 용해됨), 메탄올(약간 용해됨)
- 용해도
- 클로로포름(약간), 에틸아세테이트(약간), 메탄올(약간)에 용해됨
- 물리적 상태
- Solid
- 색상
- White to off-white
- 수용성
- (μg/L):
350 at 25–29 °C (Park and Bruce, 1968)
275 at 25 °C (quoted, Warner et al., 1987)
- Henry's Law Constant
- 0.59(x 10-5 atm?m3/mol) at 5 °C, 0.84 at 15 °C, 1.48 at 20 °C, 2.27 at 25 °C, 3.26 at 35 °C:in 3% NaCl solution: 2.07 at 5 °C, 4.93 at 15 °C, 7.70 at 25 °C, 9.28 at 35 °C (gas stripping-GC, Cetin et al., 2006)
- 노출 한도
- ACGIH TLV: TWA 0.05 mg/m3 (adopted).
- 안정성
- 감광성
안전
- 위험 및 안전 성명
- 위험 및 사전주의 사항 (GHS)
| 위험품 표기 | T,N,F | ||
|---|---|---|---|
| 위험 카페고리 넘버 | 25-33-40-50/53-39/23/24/25-23/24/25-11-52/53 | ||
| 안전지침서 | 36/37-45-60-61-16-7 | ||
| 유엔번호(UN No.) | 2761 | ||
| WGK 독일 | 3 | ||
| RTECS 번호 | PB9450000 | ||
| 위험 등급 | 6.1(a) | ||
| 포장분류 | II | ||
| 유해 물질 데이터 | 1024-57-3(Hazardous Substances Data) | ||
| 독성 | Acute oral LD50 for rats 47 mg/kg (RTECS, 1985) |
| 그림문자(GHS): |
  
|
|||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 신호 어: | Danger | |||||||||||||||||||||||||||||||||||
| 유해·위험 문구: |
|
|||||||||||||||||||||||||||||||||||
| 예방조치문구: |
|
헵타클로르 에폭시드 C화학적 특성, 용도, 생산
용도
The cis-metabolite of organochlorine pesticide Heptachlor.정의
A degradation product of heptachlor that also acts as an insecticide.일반 설명
Heptachlor epoxide is also a white powder. Bacteria and animals break down heptachlor to form heptachlor epoxide. The epoxide is more likely to be found in the environment than heptachlor. Heptachlor epoxide is a degradation product of heptachlor that occurs in soil and in or on crops when treatments with heptachlor, an insecticide, have been made. It forms readily upon exposing heptachlor to air. The U.S. EPA lists heptachlor epoxide as a possible human carcinogen.반응 프로필
CIS-HEPTACHLOREPOXIDE EXO-, ISOMER B may react with acids, bases, and oxidizing and reducing agents.위험도
Possible carcinogen.건강위험
ACUTE/CHRONIC HAZARDS: Toxic.화재위험
Non-combustible, substance itself does not burn but may decompose upon heating to produce corrosive and/or toxic fumes. Containers may explode when heated. Runoff may pollute waterways.Safety Profile
Confirmed carcinogen with experimental carcinogenic data. Poison by ingestion and intravenous routes. Human mutation data reported. When heated to decomposition it emits toxic fumes of Cl-. See also HEITACHLOR환경귀착
Biological. In a model ecosystem containing plankton, Daphnia magna, mosquito larva (Culex pipiens quinquefasciatus), ?sh (Cambusia af?nis), alga (Oedogonium cardiacum) and snail (Physa sp.), heptachlor epoxide degraded to hydroxychlordene epoxide (Lu et al., 1975). Using settled domestic wastewater inoculum, heptachlor epoxide (5 and 10 mg/L) did not degrade after 28 days of incubation at 25°C (Tabak et al., 1981). This is consistent with the ?ndings of Bowman et al. (1965). They observed that under laboratory conditions, heptachlor epoxide did not show any evidence of degradation when incubated in a variety of soils maintained at 45°C for 8 days. The soils used in this experiment included Lakeland sand, Lynchburg loamy sand, Magnolia sandy loam, Magnolia sandy clay loam, Greenville sandy clay and Susquehanna sandy clay (Bowman et al., 1965). When heptachlor epoxide was incubated in a sandy loam soil at 28°C, however, 1hydroxychlordene formed at yields of 2.8, 5.8 and 12.0% after 4, 8 and 12 weeks, respectively (Miles et al., 1971).Photolytic. Irradiation of heptachlor epoxide by a 450-W high-pressure mercury lamp gave two half-cage isomers, each containing a ketone functional group (Ivie et al., 1972). Benson et al. (1971) reported a degradation yield of 99% when an aceton
Graham et al. (1973) reported that when solid heptachlor epoxide was exposed to July sunshine for 23.2 days, 59.3% degradation was achieved. In powdered form, however, only 5 days were required for complete degradation to occur.
Chemical/Physical. Heptachlor epoxide will hydrolyze via nucleophilic attack at the epoxide moiety forming heptachlor diol which may undergo further hydrolysis forming heptachlor triol and hydrogen chloride (Kollig, 1993).
헵타클로르 에폭시드 준비 용품 및 원자재
원자재
준비 용품
헵타클로르 에폭시드 공급 업체
글로벌( 115)공급 업체
| 공급자 | 전화 | 이메일 | 국가 | 제품 수 | 이점 |
|---|---|---|---|---|---|
| Henan Fengda Chemical Co., Ltd | +86-371-86557731 +86-13613820652 |
info@fdachem.com | China | 20291 | 58 |
| Henan Tianfu Chemical Co.,Ltd. | +86-0371-55170693 +86-19937530512 |
info@tianfuchem.com | China | 21663 | 55 |
| Hubei xin bonus chemical co. LTD | 86-13657291602 |
linda@hubeijusheng.com | CHINA | 22968 | 58 |
| career henan chemical co | +86-0371-86658258 +8613203830695 |
factory@coreychem.com | China | 29821 | 58 |
| Antai Fine Chemical Technology Co.,Limited | 18503026267 |
info@antaichem.com | CHINA | 9641 | 58 |
| TargetMol Chemicals Inc. | +1-781-999-5354 +1-00000000000 |
marketing@targetmol.com | United States | 19892 | 58 |
| HANGZHOU CLAP TECHNOLOGY CO.,LTD | 86-571-88216897,88216896 13588875226 |
sales@hzclap.com | CHINA | 6313 | 58 |
| Shaanxi Dideu Medichem Co. Ltd | +86-029-89586680 +86-18192503167 |
1026@dideu.com | China | 7726 | 58 |
| LEAP CHEM CO., LTD. | +86-852-30606658 |
market18@leapchem.com | China | 24738 | 58 |
| Hangzhou MolCore BioPharmatech Co.,Ltd. | +86-057181025280; +8617767106207 |
sales@molcore.com | China | 49739 | 58 |
헵타클로르 에폭시드 관련 검색:
헵타클로르 (R)-(?)-에피클로로히드린 에피클로로하이드린 시클로옥탄올 헵타클로르 에폭시드
3A,4,7,7A-TETRAHYDROINDENE
Heptachlor- endo- epoxide(trans-,isomer A)
CYCLOOCTENE OXIDE
BIS(4-CHLOROBUTYL) ETHER
8-Chloro-1-octanol
(R)-5-CHLORO-2-PENTANOL
6-Chlorohexanol
1,9-DICHLORONONANE
1-Chloro-4-propoxybutane
VINYLCYCLOOCTANE
2,8-Dichlorooct-1-ene
(R)-6-CHLORO-2-HEXANOL
1,1-DICHLOROCYCLOPENTANE