|
|
| | 3,5-Dibenzyloxyacetophenone Basic information |
| Product Name: | 3,5-Dibenzyloxyacetophenone | | Synonyms: | 3',5'-DIBENZYLOXYACETOPHENONE;3,5-DIBENZYLOXYACETOPHENONE;3',5'-DIBENZYLOXYACETPHENONE;3',5'-BIS(BENZYLOXY)ACETOPHENONE;1-[3,5-bis(phenylmethoxy)phenyl]-ethanon;3',5'-Dibenzyloxyacetophenone 3,5-Dibenzyloxyacetophenone;(3,5-BIS-BENZYLOXY-PHENYL)-METHANOL;Terbutaline3,5-DibenzylOxyAcetophenone | | CAS: | 28924-21-2 | | MF: | C22H20O3 | | MW: | 332.39 | | EINECS: | 249-315-7 | | Product Categories: | Aromatic Acetophenones & Derivatives (substituted);API intermediates;Building Blocks for Dendrimers;Functional Materials;Aromatics;C15 to C38;Carbonyl Compounds;Ketones;bc0001;28924-21-2 | | Mol File: | 28924-21-2.mol | 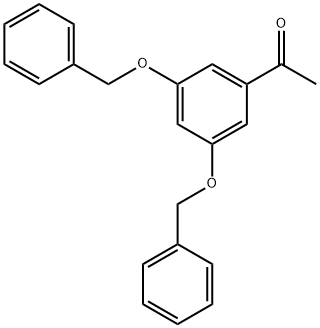 |
| | 3,5-Dibenzyloxyacetophenone Chemical Properties |
| Melting point | 60-62 °C (lit.) | | Boiling point | 429.52°C (rough estimate) | | density | 1.1079 (rough estimate) | | refractive index | 1.6000 (estimate) | | storage temp. | Sealed in dry,Room Temperature | | solubility | Soluble in dichloromethane, ethyl acetate and methanol | | form | Solid | | color | Off-White to Pale Yellow | | InChIKey | KOJXGMJOTRYLBD-UHFFFAOYSA-N | | CAS DataBase Reference | 28924-21-2(CAS DataBase Reference) | | EPA Substance Registry System | Ethanone, 1-[3,5-bis(phenylmethoxy)phenyl]- (28924-21-2) |
| Hazard Codes | Xi | | Safety Statements | 24/25 | | WGK Germany | 3 | | Hazard Note | Irritant | | TSCA | Yes | | HS Code | 29145090 |
| | 3,5-Dibenzyloxyacetophenone Usage And Synthesis |
| Chemical Properties | beige crystalline powder. | | Uses | The product 3,5-dibenzyloxyacetophenone is widely used as intermediate in the production of a pharmaceutical for treatment of obesity, diabetes, glaucoma, AIDS, HIV infection, antiinflammatory effects, and also in the treatment of disorders which result from leukotriene, tranquilizers, hypotensives, diuretics, and antiglaucoma agents.[1] | | Uses | 3,5-Dibenzyloxyacetophenone is used in the synthesis of enantiomerically pure B-ring modified analogs of (-)-epicatechin gallate. | | References | [1] Ganapati D. Yadav, Omprakash V. Badure. “Selective acylation of 1,3-dibenzyloxybenzene to 3,5-dibenzyloxyacetophenone over cesium modified dodecatungstophosphoric acid (DTP) on clay.” Applied Catalysis A: General 348 1 (2008): Pages 16-25. |
| | 3,5-Dibenzyloxyacetophenone Preparation Products And Raw materials |
| Raw materials | Ethanone, 1-[3,5-bis(phenylmethoxy)phenyl]-, oxime-->Ethanone, 1-[3,5-bis(phenylmethoxy)phenyl]-2-(methylsulfonyl)--->3,5-DIBENZYLOXYBENZOIC ACID-->3,5-Bis(benzyloxy)benzoic acid benzyl ester-->Benzyl chloride-->Benzyl bromide | | Preparation Products | Benzenemethanol, α-(bromomethyl)-3,5-bis(phenylmethoxy)-, (αS)--->3,5-Dihydroxyacetophenone-->1-Chloro-3-ethoxypropane-->5-(1-Hydroxy-2-tert-butylamino-ethyl)benzene-1,3-diol |
|