|
|
| | 3-(cyclopropylmethoxy)-4-(difluoromethoxy)benzoic Acid Basic information |
| | 3-(cyclopropylmethoxy)-4-(difluoromethoxy)benzoic Acid Chemical Properties |
| Melting point | 118-120℃ | | Boiling point | 356.4±37.0 °C(Predicted) | | density | 1.355±0.06 g/cm3(Predicted) | | storage temp. | Sealed in dry,Room Temperature | | solubility | DMSO (Slightly), Methanol (Slightly) | | form | Solid | | pka | 3.87±0.10(Predicted) | | color | White to Off-White | | InChI | InChI=1S/C12H12F2O4/c13-12(14)18-9-4-3-8(11(15)16)5-10(9)17-6-7-1-2-7/h3-5,7,12H,1-2,6H2,(H,15,16) | | InChIKey | IGFDIFLMMLWKKY-UHFFFAOYSA-N | | SMILES | C(O)(=O)C1=CC=C(OC(F)F)C(OCC2CC2)=C1 | | CAS DataBase Reference | 162401-62-9 |
| Hazard Codes | T | | Risk Statements | 25 | | Safety Statements | 45 | | RIDADR | 2811 | | HazardClass | 6.1 | | PackingGroup | Ⅲ | | HS Code | 2918999090 |
| | 3-(cyclopropylmethoxy)-4-(difluoromethoxy)benzoic Acid Usage And Synthesis |
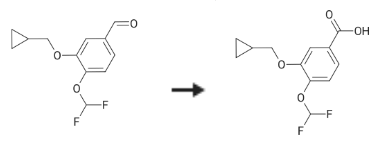
| Description | 3-(cyclopropylmethoxy)-4-(difluoromethoxy)benzoic Acid (DGM) is a pharmaceutical intermediate ingredient used in the preparation of Roflumilast, a selective phosphodiesterase-4 (PDE4) inhibitor approved for the treatment of chronic bronchitis and to reduce the risk of exacerbations in patients with severe chronic obstructive pulmonary disease (COPD). | | Uses | An impurity and intermediate in the preparation of selective phosphodiesterase 4 (PDE4) inhibitor, Roflumilast (R639700). | | Biological Activity | It was shown that 3-(cyclopropylmethoxy)-4-(difluoromethoxy)benzoic Acid (DGM) attenuated transforming growth factor-β1 (TGF-β1)-induced EMT in A549 cells and bleomycin-induced pulmonary fibrosis in rats. DGM treatment suppressed the expression of proteins such as α-SMA, waveform protein and collagen I and other proteins and increased the expression of E-cadherin. Correspondingly, the phosphorylation level of Smad2/3 was significantly reduced after DGM treatment[1]. | | Synthesis | 3-(cyclopropylmethoxy)-4-(difluoromethoxy)benzoic Acid is synthesised using 3-cyclopropylmethoxy-4-difluoromethoxybenzaldehyde as raw material by chemical reaction. The specific synthesis steps are as follows:
3-cyclopropylmethoxy-4-difluoromethoxybenzaldehyde (12 g, 50 mmol) and sulfamic acid (7.3 g, 75 mmol) were dissolved in glacial acetic acid (50 ml) and the solution added with a solution of sodium chlorite (8.2 g, 75 mmol) in water (15 ml).
The reaction mixture was stirred at room temperature for 1 hr then water (300 ml) was added so obtaining the precipitation of a solid that was filtered and dried at 40° C. under vacuum (12 g, 48 mmol, 97percent yield).
 | | References | [1] TIANXIAO SUN. Inhibitory Effects of 3-Cyclopropylmethoxy-4-(difluoromethoxy) Benzoic Acid on TGF-β1-Induced Epithelial-Mesenchymal Transformation of In Vitro and Bleomycin-Induced Pulmonary Fibrosis In Vivo.[J]. International Journal of Molecular Sciences, 2023. DOI:10.3390/ijms24076172. |
| | 3-(cyclopropylmethoxy)-4-(difluoromethoxy)benzoic Acid Preparation Products And Raw materials |
|