TriMethyl(broModifluoroMethyl)silane manufacturers
|
| | TriMethyl(broModifluoroMethyl)silane Basic information |
| Product Name: | TriMethyl(broModifluoroMethyl)silane | | Synonyms: | (Bromodifluoromethyl)trimethylsilane 98%;TriMethyl(broModifluoroMethyl)silane;Bromodifluoro(trimethylsilyl)methane;(Bromodifluoromethyl);Trimethyl(bromodifluoromethyl)silane 97%;(Bromodifluoromethyl)trimethylsilane >;Silane, (bromodifluoromethyl)trimethyl-;Difluorobromomethylsilane | | CAS: | 115262-01-6 | | MF: | C4H9BrF2Si | | MW: | 203.1 | | EINECS: | 806-938-4 | | Product Categories: | | | Mol File: | 115262-01-6.mol | 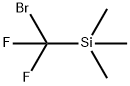 |
| | TriMethyl(broModifluoroMethyl)silane Chemical Properties |
| Boiling point | 108°C(lit.) | | density | 1.306 | | refractive index | n/D1.407 | | Fp | 46℃ | | storage temp. | -20°C | | form | liquid | | color | clear | | InChIKey | WDZVWBWAUSUTTO-UHFFFAOYSA-N | | CAS DataBase Reference | 115262-01-6 |
| Risk Statements | 11 | | Safety Statements | 24/25 | | RIDADR | UN 1993C 3 / PGIII | | TSCA | No | | HazardClass | 3 | | PackingGroup | II | | HS Code | 29319090 |
| | TriMethyl(broModifluoroMethyl)silane Usage And Synthesis |
| Chemical Properties | Colorless transparent liquid, initial boiling point 108℃, relative density 1.31 g/cm3. | | Uses | Novel difluorocarbene source for the efficient difluoromethylenation of alkenes/alkynes and difluoromethylation of O-, S-, and N-nucleophiles. | | Uses | Trimethyl(bromodifluoromethyl)silane can be used as neuroactive steroids. | | Uses | As reported by Hu and co-workers, (Bromodifluoromethyl)trimethylsilane is a highly useful reagent for the formation of difluoromethene-containing 3-membered rings and can also difluoromethylate heteroatoms with the assistance of alkaline bases, most effectively KOH. | | Application | - TriMethyl(broModifluoroMethyl)silane commonly can be used as a source for generating difluorocarbene, it is a general reagent with broad synthetic utility.
- Can generate difluorocarbene under neutral/acidic/basic conditions in the presence/absence of water at low/high temperatures.
- Can be used to prepare gem-difluorocyclopropan(e)nes, O-, S-, N-, and P-difluoromethylated compounds
| | Reactions |
(1) Difluoromethylenation of TMSCN.
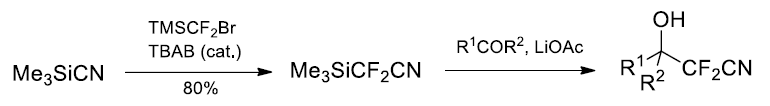
Ref. J. Org. Chem. 2012, 77, 5850?5855.
(2) Difluoromethylenation of benzyl and alkylzinc halides.

Ref. Org. Lett. 2013, 15, 917 – 919.
(3) Fluorination aminocarbonylation of aldehydes
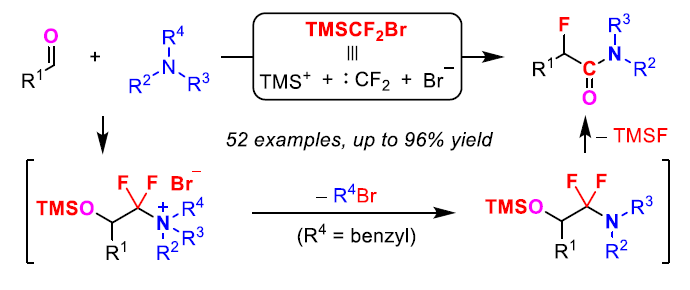
Ref. Angew. Chem. Int. Ed. 2022, e202115467
| | General Description | (Bromodifluoromethyl)trimethylsilane (TMSCF2Br) is commonly used as a source to generate dilfluorocarbene. TMSCF3 (Ruppert–Prakash reagent) and BBr3 undergoes fast halogen–exchange reaction to form MSCF2Br. | | References |
[1]Angewandte Chemie International Edition.DOI :10.1002/anie.202283811
[2]The Journal of Organic Chemistry.DOI :10.1021/acs.orglett.2c02899
|
| | TriMethyl(broModifluoroMethyl)silane Preparation Products And Raw materials |
|