- 4-Hydroxycoumarin
-

- $0.00 / 25KG
-
2024-09-30
- CAS:1076-38-6
- Min. Order: 1KG
- Purity: 99%
- Supply Ability: 100mt/year
- 4-Hydroxycoumarin
-

- $100.00/ g
-
2024-09-30
- CAS:1076-38-6
- Min. Order: 1g
- Purity: 99%
- Supply Ability: 5000
- 4-Hydroxycoumarin
-

- $10.00 / 1kg
-
2024-09-29
- CAS:1076-38-6
- Min. Order: 1kg
- Purity: 99%
- Supply Ability: 10000Ton
|
| | 4-Hydroxycoumarin Basic information |
| | 4-Hydroxycoumarin Chemical Properties |
| Melting point | 211-213 °C (lit.) | | Boiling point | 228.82°C (rough estimate) | | density | 1.1734 (rough estimate) | | vapor pressure | 0.02Pa at 25℃ | | refractive index | 1.4500 (estimate) | | storage temp. | Store below +30°C. | | solubility | DMSO (Slightly), Methanol (Slightly) | | form | Liquid | | pka | 4.50±1.00(Predicted) | | color | Clear colorless to slightly yellow | | Odor | Very faint woody-medicinal odor with a with Aluminum chloride. nut-like, hcrbaccous tone | | Water Solubility | PRACTICALLY INSOLUBLE | | BRN | 129768 | | InChIKey | VXIXUWQIVKSKSA-UHFFFAOYSA-N | | LogP | 1.6-2.31 at 21.2℃ | | CAS DataBase Reference | 1076-38-6(CAS DataBase Reference) | | NIST Chemistry Reference | 4-Hydroxycoumarin(1076-38-6) | | EPA Substance Registry System | 2H-1-Benzopyran-2-one, 4-hydroxy- (1076-38-6) |
| Hazard Codes | Xn,Xi | | Risk Statements | 22-36/37/38 | | Safety Statements | 26-36 | | WGK Germany | 3 | | RTECS | DJ3100000 | | Hazard Note | Irritant | | TSCA | Yes | | HS Code | 29322980 |
| | 4-Hydroxycoumarin Usage And Synthesis |
| Chemical Properties | Yellow powder or chunks or Slightly yellow needle-like crystals. freely soluble in ethanol, ether and hot water. brown in color with ferric chloride. | | Uses | 4-Hydroxycoumarin may be regarded as a phenylenetetronic acid , and like tetronic acid forms condensation products with aldehydes.
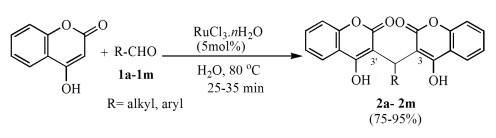
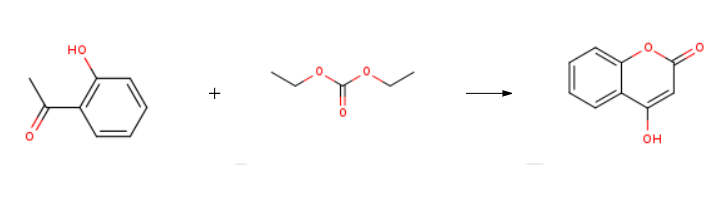
Synthesis of biscoumarin derivatives by the reaction of aldehydes and 4-hydroxycoumarin using ruthenium(III) chloride hydrate as a versatile homogeneous catalyst. | | Uses | 4-Hydroxycoumarin is involved in annulation reactions, due to the relatively high acidity of the C-H bond at the 3-position: a three-component reaction with isocyanides and dialkyl acetylene dicarboxylates affords annulated 4H-pyrans. | | Uses | 4-Hydroxycoumarin is a plant derived antioxidant, protecting against lipid peroxidation, as well as a potential inhibitor of HIV-1 Integrase. 4-Hydroxycoumarins are a very important class of biologically active drugs which are widely used as anticoagulants-Warfarin and Acenocoumarol. | | Definition | ChEBI: 4-hydroxycoumarin is a hydroxycoumarin that is coumarin in which the hydrogen at position 4 is replaced by a hydroxy group. It is a conjugate acid of a 4-hydroxycoumarin(1-). | | Synthesis Reference(s) | The Journal of Organic Chemistry, 25, p. 677, 1960 DOI: 10.1021/jo01074a630 | | Flammability and Explosibility | Not classified | | Synthesis | 4-Hydroxycoumarin is prepared by the reaction of o-hydroxyacetophenone and Diethyl carbonate. The steps are as follows:
General procedure: To a stirred mixture (1.15 mmol) and sodium hydride (138 mg, 5.75 mmol) in 5 ml dry toluene, a solution of the diethyl carbonate (1.15 mmol) in dry toluene was dropped. The mixture was reacted at 0°C for 30 min, then heated to reflux and reacted for another 4 h. The reaction mixture was quenched with water (20 mL) at ice bath and neutralized to pH 7 with 2 N HCl. The precipitate was filtered, washed with cold water and dried to get 6a-f. 1.2.1.1. 4-Hydroxy-2H-chromen-2-one (6a) White solidyield: 95; 1H NMR (600 MHz, DMSO-d6) δ 12.53 (s, 1H), 7.83 (d, J = 7.2 Hz, 1H), 7.65 (d, J = 7.8 Hz, 1H), 7.38-7.34 (m, 2H), 5.60 (s, 1H).
 | | Purification Methods | Crystallise 4-hydroxycoumarin from water and dry it in a vacuum desiccator over pKEst Sicapent. [Beilstein 18/1 V 378.] |
| | 4-Hydroxycoumarin Preparation Products And Raw materials |
|