|
|
| | 2-amino-1-(4-chlorobenzyl)-5-trifluoromethylbenzimidazole Basic information |
| Product Name: | 2-amino-1-(4-chlorobenzyl)-5-trifluoromethylbenzimidazole | | Synonyms: | 2-amino-1-(4-chlorobenzyl)-5-trifluoromethylbenzimidazole;NS 638;1H-Benzimidazol-2-amine,1-[(4-chlorophenyl)methyl]-5-(trifluoromethyl)-;1-[(4-Chlorophenyl)Methyl]-5-(Trifluoromethyl)Benzimidazol-2-Amine;NS-638;NS 638;NS638, >98%;1-(4-Chlorobenzyl)-5-(trifluoromethyl)-1H-benzo[d]imidazol-2-amine;1-(4-Chlorobenzyl)-5-(trifluoromethyl)-1H-benzo[d]imidazol-2-amine[NS638 | | CAS: | 150493-34-8 | | MF: | C15H11ClF3N3 | | MW: | 325.72 | | EINECS: | | | Product Categories: | | | Mol File: | 150493-34-8.mol | 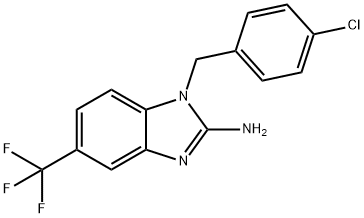 |
| | 2-amino-1-(4-chlorobenzyl)-5-trifluoromethylbenzimidazole Chemical Properties |
| Boiling point | 476.3±55.0 °C(Predicted) | | density | 1.45±0.1 g/cm3(Predicted) | | storage temp. | Store at -20°C | | solubility | DMF: 5 mg/ml; DMSO: 5 mg/ml; Ethanol: 2 mg/ml; PBS (pH 7.2): insol | | form | A solid | | pka | 5.15±0.10(Predicted) | | color | White to off-white |
| | 2-amino-1-(4-chlorobenzyl)-5-trifluoromethylbenzimidazole Usage And Synthesis |
| Biological Activity | NS-638 is a non-peptidic small molecule with Ca2+-ion channel blocking properties. The IC50 value for intracellular blocking of the increase in Ca2+ concentration activated by K+ was 3.4 μM. | | in vivo | In the mouse middle cerebral artery occlusion model, NS-638 administered ip (50 mg kg-1) at 1 h and 6 h post-ischemia, and once a day for the next two days, results in a 48% reduction in total infarct volume. It does not show protection against ischemic neuronal damage in the gerbil model of bilateral carotid artery occlusion. | | target | IC50: 3.4 μM (K + -stimulated intracellular Ca 2+ -elevation) |
| | 2-amino-1-(4-chlorobenzyl)-5-trifluoromethylbenzimidazole Preparation Products And Raw materials |
|